📋 Key Information Summary
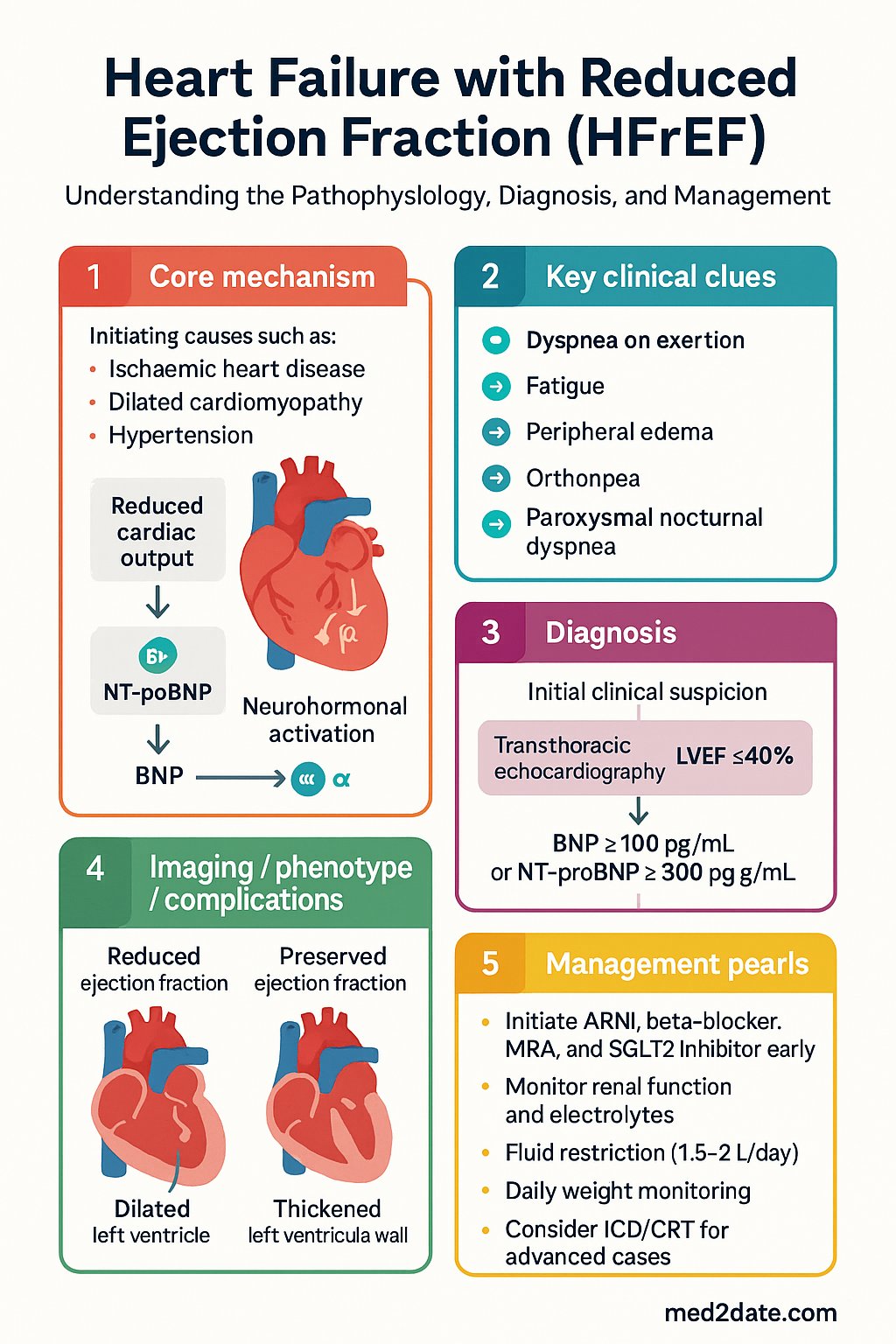
- HFrEF is defined as heart failure symptoms with LVEF ≤40% on transthoracic echocardiography; BNP ≥100 pg/mL or NT-proBNP ≥300 pg/mL supports diagnosis and correlates with disease severity.
- The four pillars of guideline-directed medical therapy (GDMT) are ARNI (or ACEi/ARB), beta-blocker, mineralocorticoid receptor antagonist (MRA), and SGLT2 inhibitor — all four should be initiated as early as tolerated, ideally within the first four weeks of diagnosis.
- Sacubitril–valsartan (Entresto®) is PBS-listed as a Restricted Benefit for NYHA II–IV HFrEF and should replace ACEi/ARB when tolerated; it must not be used within 36 hours of an ACE inhibitor dose.
- Three evidence-based beta-blockers reduce mortality in HFrEF: carvedilol (Dilatrend®), bisoprolol (not PBS-listed for HF), and metoprolol succinate (Betaloc CR®); start low and titrate every 2–4 weeks to maximum tolerated dose.
- SGLT2 inhibitors (dapagliflozin 10 mg PO daily or empagliflozin 10 mg PO daily) are effective regardless of diabetes status; they are PBS-listed for HFrEF and should be initiated early in the GDMT pathway.
- ICD implantation for primary prevention is indicated when LVEF remains ≤35% despite ≥3 months of optimised GDMT, NYHA II–III, and life expectancy >1 year; CRT (cardiac resynchronisation therapy) is indicated for LVEF ≤35% with QRS ≥150 ms (LBBB morphology preferred).
- Patients with persistent NYHA III–IV symptoms despite maximal GDMT should be referred for advanced heart failure assessment including INTERMACS profiling and LVAD/transplant evaluation.
- Fluid restriction (1.5–2 L/day) and daily weight monitoring are core non-pharmacological measures; a weight gain of >1.5 kg in 24 hours or >2 kg in a week warrants medical review for diuretic adjustment.
- Aboriginal and Torres Strait Islander Australians experience HF at 1.5–2.5 times the rate of non-Indigenous Australians, with earlier onset and higher in-hospital mortality; culturally safe models of care, Telehealth access, and ACCHO-based follow-up improve outcomes.
- Up-titration of GDMT requires regular review (every 1–2 weeks initially); renal function and electrolytes should be checked within 1–2 weeks of each dose change and within 2 weeks of hospital discharge.
- Palliative care should be discussed early and integrated alongside active HF management for patients with refractory symptoms, frequent hospitalisations, or those declining advanced therapies.
Introduction & Australian Epidemiology
Heart failure with reduced ejection fraction (HFrEF) is a progressive clinical syndrome characterised by the heart's inability to pump blood effectively, resulting in elevated filling pressures, reduced cardiac output, and neurohormonal activation. HFrEF is specifically defined as heart failure with a left ventricular ejection fraction (LVEF) ≤40%, distinguishing it from heart failure with preserved ejection fraction (HFpEF, LVEF ≥50%) and heart failure with mildly reduced ejection fraction (HFmrEF, LVEF 41–49%).
In Australia, approximately 480,000 people live with heart failure, and this number is projected to exceed 600,000 by 2030 due to an ageing population and improving survival from acute cardiac events. Heart failure accounts for over 65,000 hospitalisations annually, with an average length of stay of 6–7 days, making it one of the leading causes of preventable hospitalisation under the National Healthcare Agreement benchmarks.
The Australian Institute of Health and Welfare (AIHW) reports that heart failure is the underlying or associated cause of death in approximately 10,000 Australians per year. The condition disproportionately affects Aboriginal and Torres Strait Islander Australians, people in rural and remote areas, and those of lower socioeconomic status. The National Heart Foundation of Australia and the Cardiac Society of Australia and New Zealand (CSANZ) jointly publish clinical guidelines that align broadly with the European Society of Cardiology (ESC) 2021 and American Heart Association (AHA)/American College of Cardiology (ACC) 2022 frameworks, adapted for Australian practice.
Key aetiologies of HFrEF in the Australian context include ischaemic heart disease (the most common cause, accounting for approximately 60–70% of cases), dilated cardiomyopathy (idiopathic, familial, or toxin-related including alcohol), uncontrolled hypertension, valvular heart disease, myocarditis, peripartum cardiomyopathy, and tachycardia-mediated cardiomyopathy. Iron deficiency (with or without anaemia) is present in up to 50% of HFrEF patients and independently worsens outcomes.
Diagnostic Criteria
Echocardiographic Assessment
Transthoracic echocardiography (TTE) is the cornerstone of HFrEF diagnosis and should be performed in all patients with suspected heart failure. The diagnosis of HFrEF requires:
- LVEF ≤40% measured by modified Simpson's biplane method (preferred) or other validated quantitative technique
- Assessment of left ventricular dimensions (LVEDD, LVESD) and wall thickness
- Diastolic function grading (E/e' ratio, left atrial volume index, tricuspid regurgitation velocity)
- Evaluation for regional wall motion abnormalities suggesting ischaemic aetiology
- Assessment of right ventricular function (TAPSE, RV S') and pulmonary artery systolic pressure
- Valvular assessment for underlying structural causes or secondary regurgitation
- Estimation of filling pressures and volume status
A repeat echocardiogram should be performed 3–6 months after initiating GDMT to assess treatment response, and again if there is a significant clinical change (e.g., new murmur, clinical deterioration, or before considering device therapy). An LVEF improvement to >40% (HFimpEF) does not remove the diagnosis of HFrEF; patients should continue all GDMT indefinitely as cardiac remodelling may reverse with treatment withdrawal.
BNP / NT-proBNP Interpretation
Natriuretic peptides are essential for both diagnosis and prognostication. Australian laboratories routinely offer both BNP and NT-proBNP. Results should be interpreted in the clinical context, noting that obesity, atrial fibrillation, renal function, and age significantly influence levels.
| Biomarker | Exclusion Threshold (rule out HF) | Diagnosis Favourable | Notes |
|---|---|---|---|
| BNP | <100 pg/mL | ≥100 pg/mL | Less affected by age and renal function than NT-proBNP; affected by neprilysin inhibitors (sacubitril) |
| NT-proBNP | <300 pg/mL | ≥300 pg/mL | Higher thresholds in older adults; renal impairment raises levels independent of HF |
| NT-proBNP (age-adjusted) | <125 pg/mL (<75 years); <450 pg/mL (≥75 years) | Above age-specific thresholds | ESC 2021 age-adjusted cut-offs improve specificity in elderly patients |
Aetiology Determination
Identifying the underlying cause of HFrEF is essential for guiding specific therapy and prognosis. A structured approach to aetiology determination should include:
| Investigation | Purpose | Availability / MBS |
|---|---|---|
| Coronary angiography (invasive or CTCA) | Exclude ischaemic aetiology; consider if angina, regional wall motion abnormalities, or ischaemic risk factors present | Available in all tertiary centres; MBS items for CT coronary angiography and invasive angiography |
| Cardiac MRI (CMR) | Tissue characterisation (oedema, fibrosis, infiltration); differentiate ischaemic vs non-ischaemic; detect myocarditis, sarcoidosis, ARVC | Available at major metropolitan centres; MBS item 63448 (cardiac MRI with contrast) |
| Iron studies (ferritin, transferrin saturation) | Iron deficiency assessment — ferritin <100 µg/L or 100–299 µg/L with transferrin saturation <20% | Widely available; MBS item 66591 (iron studies) |
| Thyroid function tests | Exclude thyroid dysfunction as reversible cause | Widely available; MBS item 66719 |
| FBC, renal function, LFTs, glucose, lipids | Baseline organ function, comorbidity screening | Widely available |
| Genetic testing / family screening | Suspected familial dilated cardiomyopathy (≥2 affected family members, young onset, conduction disease, skeletal myopathy) | Referral to genetics service; available through state genetics services |
| Endomyocardial biopsy | Suspected giant cell myocarditis, eosinophilic myocarditis, cardiac amyloidosis (when non-invasive testing inconclusive), cardiac transplant rejection | Tertiary centre referral only; limited availability |
| 99mTc-DPD / PYP scintigraphy | Transthyretin cardiac amyloidosis screening | Available at selected nuclear medicine centres; MBS item 61323 |
| Sleep study (polysomnography) | Screen for obstructive sleep apnoea, a common comorbidity that worsens HF outcomes | Available in sleep laboratories and home-based testing; MBS item 12203 / 12250 |
All patients should have a comprehensive history and examination to identify potential reversible and treatable causes including alcohol excess, recreational drug use (cocaine, methamphetamine), chemotherapy exposure (anthracyclines, trastuzumab), autoimmune conditions, and infective aetiologies (Chagas disease in relevant populations).
Guideline-Directed Medical Therapy (GDMT)
Guideline-directed medical therapy for HFrEF has evolved into a four-pillar approach based on landmark randomised controlled trials demonstrating mortality and hospitalisation benefits. The four pillars are: (1) ARNI (or ACEi/ARB), (2) beta-blocker, (3) mineralocorticoid receptor antagonist (MRA), and (4) SGLT2 inhibitor. All four should be initiated in hospitalised patients before discharge where feasible and in outpatients as early as possible after diagnosis.
Pillar 1: ARNI / ACEi / ARB
Sacubitril–valsartan (Entresto®), a combined neprilysin inhibitor and ARB, is the preferred first-line agent based on the PARADIGM-HF trial (20% relative risk reduction in cardiovascular death or HF hospitalisation vs enalapril). It is PBS-listed as a Restricted Benefit for chronic heart failure (NYHA II–IV) with LVEF ≤40% on optimised background therapy. An ACEi or ARB remains appropriate when ARNI is not tolerated or not accessible.
Pillar 2: Beta-Blockers
Only three beta-blockers have demonstrated mortality benefit in HFrEF: carvedilol (COPERNICUS, US Carvedilol HF trials), bisoprolol (CIBIS-II), and metoprolol succinate (MERIT-HF). Other beta-blockers, including atenolol and metoprolol tartrate, should not be used for this indication. Beta-blockers should be initiated at low dose once the patient is euvolaemic and haemodynamically stable, then titrated upward every 2–4 weeks to maximum tolerated dose.
Pillar 3: Mineralocorticoid Receptor Antagonists (MRAs)
Spironolactone and eplerenone reduce mortality and hospitalisation in HFrEF (RALES, EMPHASIS-HF trials). MRAs should be initiated once serum potassium is <5.0 mmol/L and eGFR is ≥30 mL/min/1.73m². Hyperkalaemia and worsening renal function are the most common adverse effects requiring monitoring.
Pillar 4: SGLT2 Inhibitors
SGLT2 inhibitors (dapagliflozin and empagliflozin) have demonstrated robust reductions in the composite of cardiovascular death or HF hospitalisation in the DAPA-HF and EMPEROR-Reduced trials, respectively. Benefits were consistent regardless of diabetes status, and these agents should be considered foundational therapy in all HFrEF patients. They are now PBS-listed for HFrEF independently of diabetes.
Diuretics for Volume Management
Loop diuretics (furosemide, bumetanide) are essential for managing congestion but do not improve mortality. They should be prescribed at the lowest dose that maintains euvolaemia. In patients with diuretic resistance, sequential nephron blockade with the addition of metolazone (2.5–5 mg PO daily/alternate days) or thiazide diuretic is recommended, with close electrolyte monitoring.
Additional Pharmacotherapy
For patients who remain symptomatic on optimised four-pillar GDMT, additional agents to consider include:
- Ivabradine (Coralan®): Indicated when HR ≥70 bpm despite maximally tolerated beta-blocker (or if beta-blocker contraindicated), LVEF ≤35%, and in sinus rhythm. Dose: 5 mg BD, titrate to 7.5 mg BD. PBS Authority Required.
- Hydralazine + isosorbide dinitrate: Consider as add-on therapy in patients who cannot tolerate ACEi/ARB/ARNI (e.g., severe renal impairment, hyperkalaemia) or as an adjunct in self-identified African American patients (A-HeFT trial). Not commonly used in Australian practice.
- Intravenous iron (ferric carboxymaltose — Ferinject®): For iron-deficient HFrEF patients (ferritin <100 or ferritin 100–299 with TSAT <20%). The AFFIRM-AHF and IRONMAN trials demonstrated reduction in HF hospitalisation. Dose: 500–1000 mg IV per infusion, with repeat dosing guided by iron studies at 4–6 weeks. PBS-listed for iron deficiency with chronic heart failure.
- Vericiguat (Verquvo®): Oral soluble guanylate cyclase stimulator for patients with worsening HF on optimised GDMT (VICTORIA trial). Not yet PBS-listed in Australia; available via compassionate access or private script in selected cases.
Device Therapy
Implantable Cardioverter-Defibrillator (ICD) — Primary Prevention
ICD implantation for primary prevention of sudden cardiac death is indicated in patients with HFrEF who meet all of the following criteria:
- LVEF ≤35% despite ≥3 months of optimised GDMT (documented at ≥4 weeks after most recent dose change or hospitalisation)
- NYHA functional class II or III (ambulatory)
- Expected survival with good functional status >1 year
- Sinus rhythm preferred (benefit less clear in atrial fibrillation, individualised decision)
ICD — Secondary Prevention
ICD implantation is indicated for secondary prevention in survivors of cardiac arrest due to ventricular fibrillation or sustained ventricular tachycardia (VT) not due to a reversible cause, or in patients with spontaneous sustained VT with structural heart disease or haemodynamic compromise. These indications apply regardless of LVEF once the acute event has been managed.
Cardiac Resynchronisation Therapy (CRT)
CRT with biventricular pacing (CRT-P) or CRT combined with defibrillator (CRT-D) improves symptoms, reduces hospitalisation, and reduces mortality in selected patients with HFrEF and electrical dyssynchrony.
| Indication | QRS Criteria | LVEF | NYHA | Rhythm |
|---|---|---|---|---|
| Class I (strongest) | ≥150 ms, LBBB morphology | ≤35% | II–IV (ambulatory) | Sinus rhythm |
| Class IIa | ≥150 ms, LBBB | ≤35% | II–IV | Atrial fibrillation (consider AV node ablation) |
| Class IIa | 130–149 ms, LBBB | ≤35% | II–IV | Sinus rhythm |
| Class IIb | ≥150 ms, non-LBBB | ≤35% | II–IV | Sinus rhythm |
| Class III (not indicated) | <130 ms | Any | Any | Any |
CRT-D is generally preferred over CRT-P for patients who also meet primary prevention ICD criteria. CRT-P alone may be appropriate for elderly patients, those with significant comorbidities, or those who decline a defibrillator. Conduction system pacing (His bundle pacing or left bundle branch area pacing) is an emerging alternative to conventional CRT when coronary sinus lead placement is not feasible.
Wearable Cardioverter-Defibrillator (WCD)
A wearable cardioverter-defibrillator (LifeVest®) provides temporary protection against sudden cardiac death during the "window of opportunity" before ICD implantation may be indicated. Indications include:
- LVEF ≤35% within the first 40 days post-MI
- Newly diagnosed non-ischaemic cardiomyopathy within the first 3–9 months of GDMT initiation
- Bridge to transplant or LVAD (where ICD is not yet implanted)
- Patients with a temporary indication who have contraindications to implantation (e.g., active infection)
WCD availability in Australia is limited to major centres. Adherence is the primary barrier — patients must wear the device ≥20 hours/day for it to be effective. Average wear time in clinical practice is 15–18 hours/day.
Advanced Heart Failure & Transplantation
Defining Advanced Heart Failure
Advanced (or stage D) heart failure is characterised by severe symptoms (NYHA III–IV) despite maximally tolerated GDMT, device therapy where appropriate, and treatment of reversible factors. Patients typically experience recurrent hospitalisations (≥2 in 12 months for HF), worsening renal function, intolerance to GDMT, hypotension, hyponatraemia, and increasing diuretic requirements. Referral to an advanced heart failure specialist centre should not be delayed — early referral improves outcomes for patients who may benefit from transplantation or mechanical circulatory support.
INTERMACS Profiles
The Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) profiles classify advanced HF patients by acuity, guiding urgency for mechanical circulatory support and transplantation listing.
Left Ventricular Assist Device (LVAD)
Durable LVAD therapy (predominantly the HeartMate 3™ centrifugal-flow pump) is indicated as bridge to transplant (BTT) or as destination therapy (DT) for patients with advanced HF who are not transplant candidates. In Australia, LVAD implantation is performed at specialised centres in major cities (Royal Prince Alfred Hospital, St Vincent's Hospital Sydney, Alfred Hospital Melbourne, Prince Charles Hospital Brisbane, Fiona Stanley Hospital Perth).
Heart Transplantation
Cardiac transplantation remains the gold standard for advanced HFrEF, with median post-transplant survival exceeding 13 years in Australia. Indications include severe refractory HF with poor prognosis despite maximal medical and device therapy, and absence of significant contraindications.
Absolute contraindications include active malignancy (excluding low-risk skin cancers), active substance abuse, severe irreversible pulmonary hypertension (PVR >5 Wood units unresponsive to vasodilator testing), active systemic infection, and significant psychosocial barriers to adherence.
Australian transplant centres: Heart transplantation is performed at seven nationally accredited centres — St Vincent's Hospital Sydney, Royal Prince Alfred Hospital Sydney, Alfred Hospital Melbourne, Royal Melbourne Hospital, Prince Charles Hospital Brisbane, Fiona Stanley Hospital Perth, and Royal Adelaide Hospital. Approximately 100–120 heart transplants are performed annually in Australia, with wait-list times ranging from 3–12 months depending on blood group, body size, and clinical urgency (Status 1 listing for high-acuity patients).
Palliative Care in HFrEF
Palliative care should be integrated early and concurrently with active HF management, not reserved for the terminal phase. Key triggers for palliative care referral include:
- NYHA III–IV symptoms despite optimised GDMT and devices
- ≥2 HF hospitalisations in 12 months
- Declining or ineligible for advanced therapies (transplant, LVAD)
- Patient preference to focus on quality of life
- Significant non-cardiac comorbidities limiting prognosis
Advance care planning discussions, including documentation of resuscitation preferences (Advance Care Directive / substitute decision-maker designation), should be initiated at the time of device consideration or when prognosis is anticipated to be limited. Symptom management with opioids for refractory dyspnoea, anxiolytics for breathlessness-associated anxiety, and management of fluid overload with intermittent subcutaneous furosemide infusions (in community palliative care settings) should be considered. Australian palliative care services are accessible via referral through state-based palliative care networks and community palliative care teams.

Monitoring & Follow-up
Clinical Assessment Schedule
A structured, multidisciplinary approach to follow-up reduces readmissions and improves mortality. Heart failure nurse-led titration clinics and telehealth models have been shown to improve GDMT uptake in Australian settings (NHFA/CSANZ 2024 guidance).
Medication Titration Protocol
Systematic up-titration is critical — the majority of Australian HF patients are sub-optimally dosed, particularly beta-blockers and ACEi/ARB/ARNI. The following protocol applies:
| Medication | Titration Interval | Key Monitoring | Stop/Revert Criteria |
|---|---|---|---|
| ARNI / ACEi / ARB | Every 2–4 weeks | BP, K⁺, Cr within 1–2 weeks of each dose change | SBP <90 mmHg, K⁺ >5.5 mmol/L, Cr rise >30% from baseline, symptomatic hypotension |
| Beta-blockers | Every 2–4 weeks | HR (target 55–70 bpm at rest), BP, symptoms | HR <50 bpm, SBP <90, worsening HF symptoms (may need to reduce diuretic first before halting beta-blocker uptitration) |
| MRAs | Single dose step (12.5 → 25 → 50 mg) | K⁺ and Cr at 3 days, 1 week, and 1 month after initiation; then every 3–6 months | K⁺ >5.5 mmol/L, eGFR <30 mL/min/1.73m² |
| SGLT2 inhibitors | No titration (fixed dose) | eGFR at 2–4 weeks (expect transient dip); BP; symptoms of volume depletion | eGFR decline >30% sustained (unusual); symptomatic hypotension |
Remote Monitoring
Remote monitoring strategies are increasingly incorporated into Australian HF care models:
- Structured telephone support: Regular nurse-led phone calls (weekly initially, then monthly) for symptom assessment, weight review, and medication titration. Multiple Australian RCTs (e.g., TIM-HF2-equivalent programs) demonstrate reduced readmissions. Funded through state heart failure programs and Medicare chronic disease management items.
- Telehealth video consultations: Particularly valuable in rural and remote Australia where specialist access is limited. Medicare Benefits Schedule items for telehealth have been expanded post-COVID and remain available for cardiologist and GP reviews.
- Implantable haemodynamic monitoring: CardioMEMS™ (pulmonary artery pressure monitoring) is available in selected Australian centres. The CHAMPION trial demonstrated a 37% reduction in HF hospitalisation. Access is currently limited to research or compassionate use in most jurisdictions.
- Patient self-management education: Daily weight monitoring, fluid restriction adherence (typically 1.5–2 L/day), sodium restriction (<2 g/day), exercise prescription (cardiac rehabilitation), and recognition of decompensation signs (weight gain >1.5 kg in 24 hours, increasing dyspnoea, oedema).
Special Populations
Pregnancy
Contraindicated in pregnancy: ACEi, ARB, ARNI, MRA, SGLT2 inhibitors — all are teratogenic (Category D). Women of childbearing potential should receive contraception counselling when initiated on GDMT.
Peripartum cardiomyopathy: Defined as LVEF ≤45% in the last month of pregnancy or within 5 months postpartum. Management includes bromocriptine (2.5 mg PO BD for 2 weeks then 2.5 mg daily for 6 weeks — evidence from the IPAC registry and small RCTs), hydralazine, and diuretics. Recovery of LVEF occurs in ~50% of cases within 6–12 months.
Breastfeeding: ACEi/ARB (enalapril, losartan) are considered compatible with breastfeeding; ARNI and SGLT2 inhibitors lack safety data — avoid.
Anticoagulation: Warfarin is teratogenic in the first trimester; use LMWH or dose-adjusted unfractionated heparin in early pregnancy if anticoagulation required (e.g., peripartum cardiomyopathy with LVEF <30%).
Paediatrics
Aetiology differs: Paediatric HF is most commonly due to congenital heart disease, dilated cardiomyopathy (genetic or myocarditis), or acquired conditions (Kawasaki disease, rheumatic heart disease in ATSI communities).
Carvedilol: 0.05 mg/kg/dose PO BD, titrate to 0.3 mg/kg/dose BD (max 25 mg BD if >35 kg). Evidence is extrapolated from adult trials; modest benefit in paediatric dilated cardiomyopathy.
Enalapril: 0.1 mg/kg/dose PO BD, titrate to 0.25–0.5 mg/kg/dose BD (max 10 mg BD).
SGLT2 inhibitors: Not approved in patients <18 years for HF indication; ongoing trials.
Diuretics: Furosemide 0.5–2 mg/kg/dose PO/IV BD–TDS; spironolactone 0.5–1 mg/kg/day PO in divided doses.
Referral: All paediatric HF should be managed by or in consultation with a paediatric cardiologist. Mechanical circulatory support (Berlin Heart EXCOR®) is available at paediatric cardiac transplant centres in Melbourne and Sydney.
Elderly (≥75 years)
Prevalence increases with age: >10% of Australians aged ≥75 years have heart failure, often with multiple comorbidities (atrial fibrillation, CKD, COPD, frailty).
GDMT tolerability: Lower starting doses and slower titration are often necessary. Monitor closely for hypotension, falls risk, renal impairment, and electrolyte disturbances. Beta-blockers and ARNI/ACEi are still indicated but target doses may be lower.
Polypharmacy: Review all medications for potential interactions and deprescribe where appropriate. NSAIDs and certain antihistamines should be avoided.
Device therapy: ICD and CRT decisions should consider frailty, cognitive status, and patient goals. CRT-P may be preferred over CRT-D in the very elderly or frail.
Frailty assessment: Use validated tools (Clinical Frailty Scale, Fried criteria) to guide treatment intensity and care planning.
Renal Impairment
Cardiorenal syndrome: HF and CKD commonly coexist (~50% of HF patients have CKD stage 3+). Worsening renal function during GDMT initiation does not necessarily mandate discontinuation — a mild rise in creatinine (up to 30%) is acceptable and expected.
ARNI: No dose adjustment for eGFR ≥20. Use cautiously below eGFR 20; limited data.
MRA: Avoid spironolactone/eplerenone if eGFR <30 mL/min/1.73m² (hyperkalaemia risk).
SGLT2 inhibitors: Can be initiated at eGFR ≥20 for HF indication (not dialysis); HF benefit is independent of kidney function.
Diuretics: Higher doses of loop diuretics may be required; consider adding metolazone for diuretic resistance.
Dialysis patients: HFrEF management in dialysis-dependent patients is challenging; beta-blockers (particularly carvedilol) have evidence of benefit. Avoid ACEi/ARB if hyperkalaemia is recurrent. Specialist nephrology–cardiology co-management is recommended.
Hepatic Impairment
Cardiac hepatopathy: Congestive hepatopathy (passive congestion) and acute cardiogenic liver injury are common in severe HF. LFT derangement is common and does not contraindicate GDMT.
Cautions: Sacubitril–valsartan: limited data in severe hepatic impairment (Child-Pugh C); use with caution. Carvedilol: bioavailability is increased in cirrhosis — start at lowest dose. Spironolactone: used therapeutically in cirrhosis for ascites, but monitor K⁺ carefully in combined HF-liver disease.
Alcohol-related cardiomyopathy: Abstinence is essential — LVEF may recover partially or completely. Refer to alcohol and other drug services. Cardiac MRI may demonstrate mid-wall fibrosis.
Immunocompromised
Transplant recipients: Heart transplant patients require lifelong immunosuppression (typically tacrolimus, mycophenolate, prednisolone). Develop a chronic immunosuppressive cardiomyopathy in a proportion of cases — manage with standard GDMT with attention to drug interactions.
Chemotherapy-induced cardiomyopathy: Anthracyclines (doxorubicin), trastuzumab, and immune checkpoint inhibitors can cause HFrEF. Trastuzumab-related cardiomyopathy is often reversible; immune checkpoint inhibitor myocarditis requires high-dose corticosteroids and cessation of immunotherapy.
HIV-associated cardiomyopathy: Manage with standard GDMT; check for drug interactions between antiretrovirals and HF medications (particularly with boosted protease inhibitors and calcium channel blockers/beta-blockers).
Chagas disease: Consider in patients from endemic regions (Central/South America) with unexplained dilated cardiomyopathy. Benznidazole (5 mg/kg/day PO for 60 days) for chronic Chagas requires infectious disease specialist guidance; cardiac management is standard GDMT.
Aboriginal and Torres Strait Islander Health Considerations
Aboriginal and Torres Strait Islander Australians experience heart failure at a significantly higher rate than non-Indigenous Australians, with onset at a younger age (often 10–15 years earlier), more advanced disease at presentation, and higher in-hospital and post-discharge mortality. Rheumatic heart disease remains an important and preventable cause of HFrEF in many remote communities, alongside ischaemic heart disease, which accounts for the majority of HF cases.
Quick Reference — GDMT Summary
📚 References
- 1. McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42(36):3599–3726.
- 2. Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145(18):e895–e1032.
- 3. Atherton JJ, Sindone A, De Pasquale CG, et al. National Heart Foundation of Australia and Cardiac Society of Australia and New Zealand: Australian Clinical Guidelines for the Management of Heart Failure 2018. Med J Aust. 2018;209(7):325–338.
- 4. McMurray JJ, Packer M, Desai AS, et al. Angiotensin–neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014;371(11):993–1004.
- 5. McMurray JJV, Solomon SD, Inzucchi SE, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2019;381(21):1995–2008.
- 6. Packer M, Anker SD, Butler J, et al. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med. 2020;383(15):1413–1424.
- 7. Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. N Engl J Med. 1999;341(10):709–717.
- 8. Packer M, Coats AJS, Fowler MB, et al. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med. 2001;344(22):1651–1658.
- 9. Mehra MR, Uriel N, Naka Y, et al. A fully magnetically levitated left ventricular assist device — final report. N Engl J Med. 2019;380(17):1618–1627.
- 10. Abraham WT, Adamson PB, Bourge RC, et al. Wireless pulmonary artery haemodynamic monitoring in chronic heart failure: a randomised controlled trial. Lancet. 2011;377(9766):658–666.
- 11. Ponikowski P, van Veldhuisen DJ, Comin-Colet J, et al. Beneficial effects of long-term intravenous iron therapy with ferric carboxymaltose in patients with symptomatic heart failure and iron deficiency. Eur Heart J. 2015;36(11):657–668.
- 12. Australian Institute of Health and Welfare. Heart, stroke and vascular disease — Australian facts. AIHW, Canberra; 2023.
- 13. National Heart Foundation of Australia. Position Statement: SGLT2 inhibitors in heart failure. NHFA, Melbourne; 2022.
- 14. Stewart S, Wiley J, Ball J, et al. Impact of nurse-led multidisciplinary management on clinical outcomes of chronic heart failure patients: the NIL-CHF randomised controlled trial. Eur J Heart Fail. 2016;18(3):284–294.
- 15. Australian Government Department of Health. National Aboriginal and Torres Strait Islander Health Plan 2021–2031. Commonwealth of Australia; 2021.
- 16. Seferović PM, Petrie MC, Filippatos GS, et al. Type 2 diabetes mellitus and heart failure: a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2018;20(5):853–872.
- 17. Bozkurt B, Coats AJS, Tsutsui H, et al. Universal definition and classification of heart failure: a report of the Heart Failure Society of America, Heart Failure Association of the European Society of Cardiology, Japanese Heart Failure Society and Writing Committee. Eur J Heart Fail. 2021;23(3):352–380.