📋 Key Information Summary
- HFpEF is defined as heart failure with LVEF ≥50%, elevated natriuretic peptides, and evidence of structural/functional cardiac abnormalities — prevalence is rising and now accounts for approximately 50% of all heart failure cases in Australia.
- The HFA-PEFF algorithm is the preferred diagnostic pathway: Step 1 (Pre-test assessment), Step 2 (Echocardiographic and natriuretic peptide scoring), Step 3 (Functional testing with diastolic stress echo or invasive haemodynamics), Step 4 (Aetiological work-up including phenotyping).
- Elevated NT-proBNP ≥125 pg/mL or BNP ≥35 pg/mL is required as an entry criterion; however, obesity, atrial fibrillation, and renal impairment significantly affect interpretation — use age-adjusted cut-offs where applicable.
- Diastolic assessment requires integrated evaluation: E/e' ratio ≥15, left atrial volume index ≥34 mL/m², tricuspid regurgitation velocity ≥2.8 m/s, and septal e' velocity <7 cm/s or lateral e' <10 cm/s.
- Empagliflozin (Jardiance®) and dapagliflozin (Forxiga®) are first-line disease-modifying therapies in HFpEF regardless of diabetes status — EMPEROR-Preserved and DELIVER trials demonstrated significant reduction in HF hospitalisation (NNT ~31 over 2 years).
- Loop diuretics (furosemide, bumetanide) remain the cornerstone of decongestive therapy; target the lowest effective dose maintaining euvolaemia with daily weight monitoring.
- Comorbidity management is central: rate control for atrial fibrillation, target BP <130/80 mmHg for hypertension, GLP-1 receptor agonists for obesity-related HFpEF (STEP-HFpEF), and optimised glycaemic control in diabetes.
- Exercise-based cardiac rehabilitation improves exercise capacity (peak VO₂), quality of life, and functional class (NYHA) — refer all eligible patients to supervised programmes.
- HFpEF phenotyping (obesity/metabolic, atrial fibrillation-driven, vascular, right ventricular dysfunction, pulmonary, rare genetic/infiltrative) guides targeted management and is increasingly important in precision medicine.
- Aboriginal and Torres Strait Islander peoples have a disproportionately higher burden of heart failure (2.7× age-adjusted prevalence), driven by earlier onset of cardiovascular risk factors — culturally safe, community-based models of care are essential.
- Special populations require dose adjustments: SGLT2 inhibitors are not recommended in severe renal impairment (eGFR <20 mL/min/1.73 m²); caution with diuretics in elderly patients to avoid pre-renal injury; pregnancy requires multidisciplinary management.
- Regular monitoring includes clinical review every 1–3 months, repeat echocardiography at 6–12 months or with clinical deterioration, serial NT-proBNP to guide therapy, and ongoing assessment for decompensation triggers.
Introduction & Australian Epidemiology
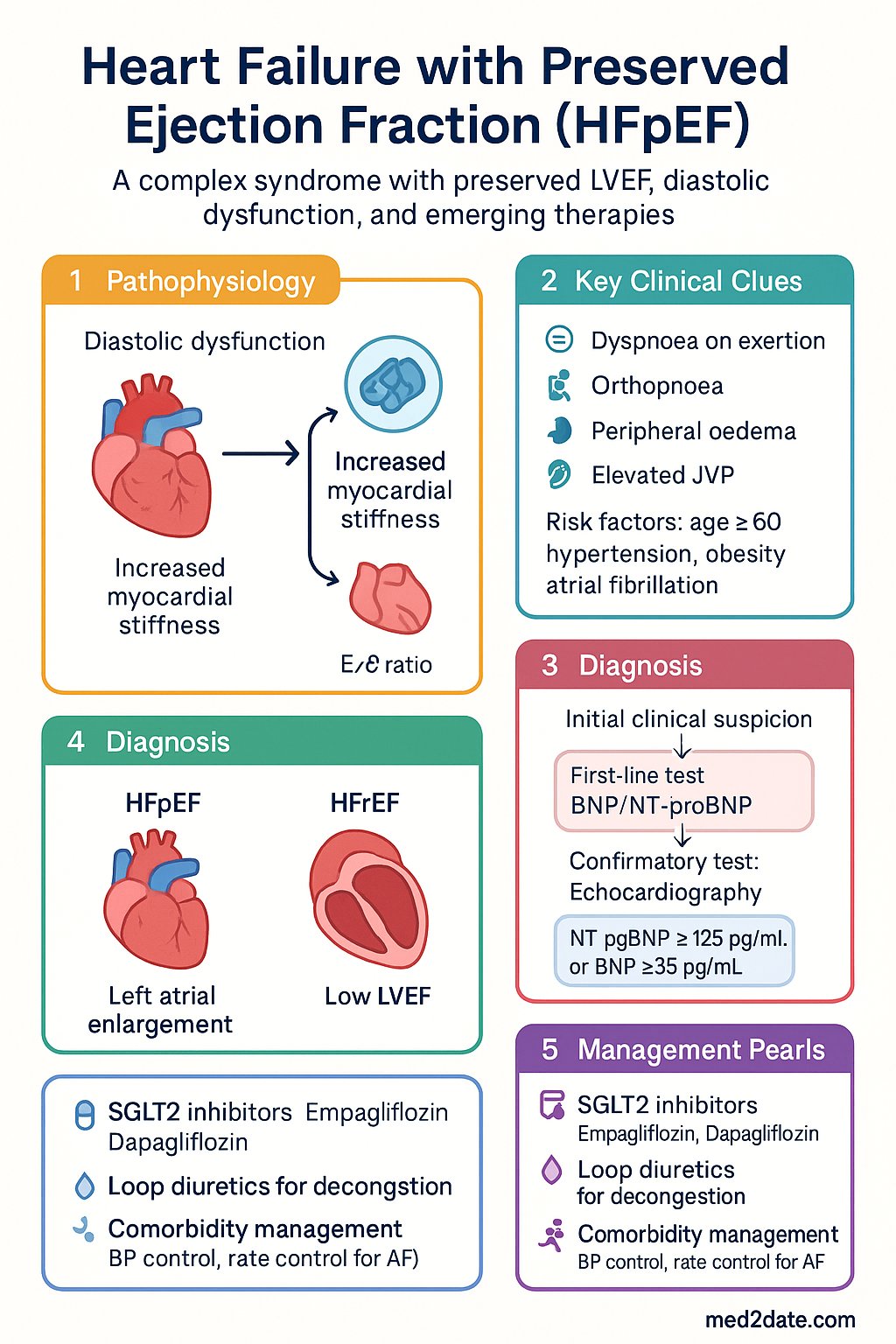
Heart failure with preserved ejection fraction (HFpEF) is a complex clinical syndrome characterised by symptoms and signs of heart failure in the presence of a left ventricular ejection fraction (LVEF) ≥50%, alongside evidence of elevated left ventricular filling pressures and structural or functional cardiac abnormalities. HFpEF represents a heterogeneous group of disorders unified by a common final pathway of diastolic dysfunction, increased myocardial stiffness, and impaired ventricular-arterial coupling.
Unlike heart failure with reduced ejection fraction (HFrEF), HFpEF has historically lacked robust evidence-based therapies, earning it the moniker of a "therapeutic desert." However, the landscape has shifted dramatically with the publication of landmark SGLT2 inhibitor trials and emerging data on GLP-1 receptor agonists, fundamentally changing the management paradigm.
Australian Burden of Disease
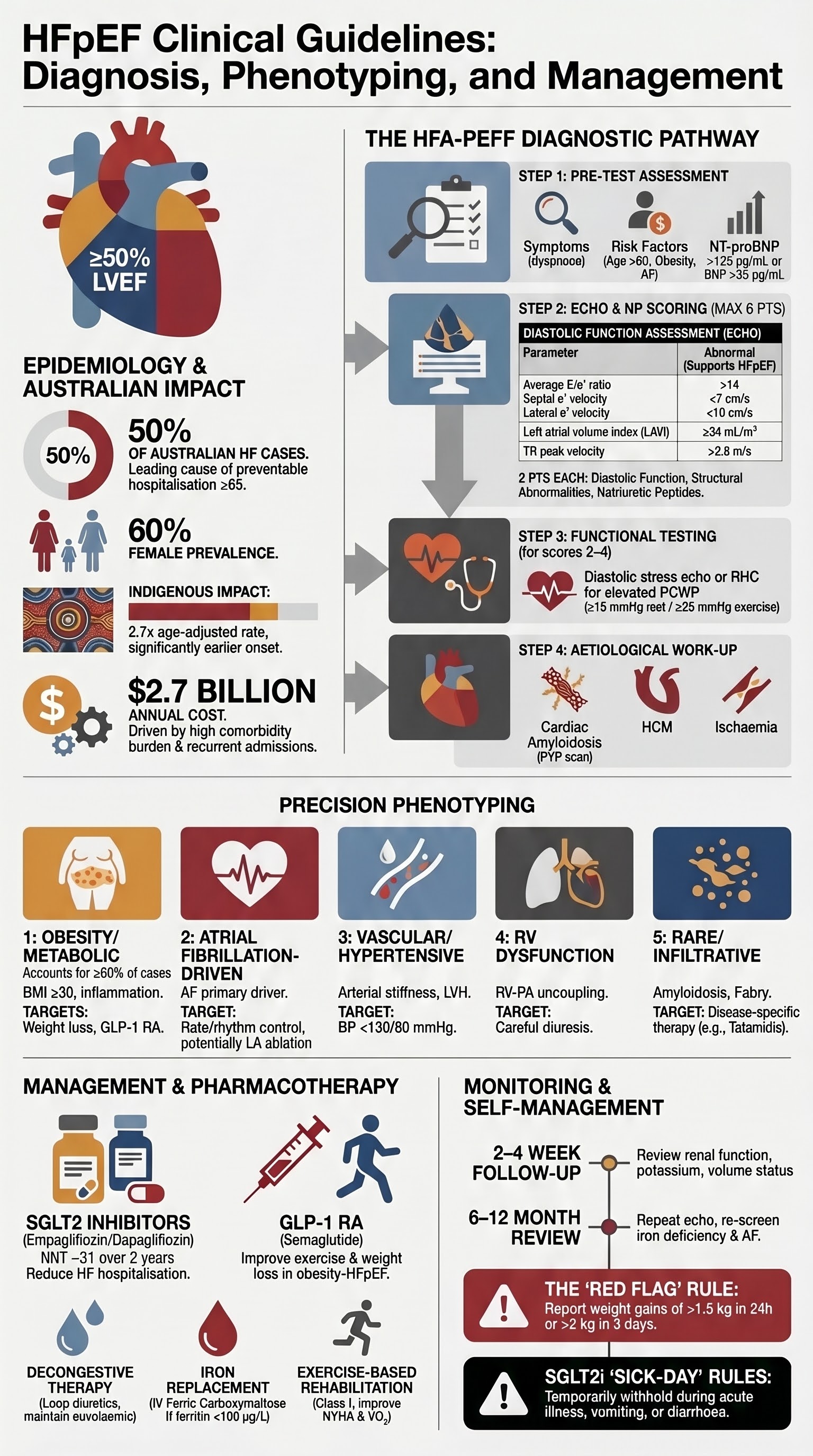
The Australian Institute of Health and Welfare (AIHW) estimates that approximately 480,000 Australians are living with heart failure, with HFpEF accounting for roughly half of all cases. The prevalence of HFpEF is increasing due to population ageing, rising rates of obesity, type 2 diabetes mellitus, hypertension, and atrial fibrillation. Key Australian epidemiological data include:
- Heart failure prevalence increases from approximately 1–2% in those aged <55 years to >10% in those aged ≥75 years.
- HFpEF accounts for approximately 50% of incident heart failure hospitalisations, with this proportion rising over the past two decades.
- In-hospital mortality for HFpEF-related admissions is approximately 4–6%, with 30-day readmission rates of 20–25%.
- Heart failure is the leading cause of preventable hospitalisation in Australians aged ≥65 years (AIHW National Hospital Morbidity Database).
- The direct cost of heart failure management in Australia is estimated at $2.7 billion annually, with HFpEF patients contributing disproportionately due to comorbidity burden and recurrent admissions.
- Women are disproportionately affected by HFpEF compared to HFrEF, representing approximately 60% of HFpEF cases.
- Aboriginal and Torres Strait Islander Australians experience heart failure at 2.7 times the age-adjusted rate of non-Indigenous Australians, with earlier onset and more severe disease at presentation.
Classification and Terminology
The 2021 ESC and 2022 AHA/ACC/HFSA guidelines classify heart failure into categories based on LVEF:
| Category | LVEF | Abbreviation | Description |
|---|---|---|---|
| Reduced | ≤40% | HFrEF | Established evidence base for neurohormonal therapy |
| Mildly reduced | 41–49% | HFmrEF | Intermediate phenotype; growing evidence base |
| Preserved | ≥50% | HFpEF | Heterogeneous; SGLT2i and GLP-1 RA now evidence-based |
| Improved | Previously ≤40%, now >40% | HFimpEF | Treated HFrEF with recovered LVEF; continue GDMT |
Diagnostic Criteria & Phenotyping
Diagnosis of HFpEF remains one of the most challenging tasks in clinical cardiology. Unlike HFrEF, where reduced LVEF provides a straightforward diagnostic anchor, HFpEF requires integration of clinical, biochemical, echocardiographic, and sometimes invasive haemodynamic data. The HFA-PEFF diagnostic algorithm, developed by the Heart Failure Association of the ESC, provides a structured, evidence-based approach.
HFA-PEFF Diagnostic Algorithm
The HFA-PEFF algorithm is a stepwise, standardised approach to HFpEF diagnosis comprising four steps:
Diastolic Function Assessment
Echocardiographic evaluation of diastolic function is central to HFpEF diagnosis. The 2016 ASE/EACVI guidelines recommend an integrated approach using multiple parameters:
| Parameter | Normal | Abnormal (supports HFpEF) | Grey Zone |
|---|---|---|---|
| Septal e' velocity | ≥7 cm/s | <7 cm/s | 7–10 cm/s |
| Lateral e' velocity | ≥10 cm/s | <10 cm/s | 10–12 cm/s |
| Average E/e' ratio | <10 | >14 | 10–14 |
| Left atrial volume index | <34 mL/m² | ≥34 mL/m² | — |
| TR peak velocity | ≤2.8 m/s | >2.8 m/s | — |
| LAVI/LAEF change with Valsalva | LAVI decreases | No decrease (loss of LA compliance reserve) | — |
Invasive Haemodynamics
Right heart catheterisation (RHC) remains the gold standard for confirming elevated filling pressures when non-invasive assessment is inconclusive. Key haemodynamic criteria include:
- Resting PCWP ≥15 mmHg (measured at end-expiration, mean) is diagnostic of elevated left-sided filling pressures.
- Exercise PCWP ≥25 mmHg during supine or upright exercise is diagnostic of HFpEF when resting pressures are normal — this identifies "occult" or "exertional" HFpEF.
- Exercise-induced elevation in pulmonary artery pressure (mPAP ≥30 mmHg) with normal resting haemodynamics is characteristic of early-stage HFpEF.
- Left ventricular end-diastolic pressure (LVEDP) ≥16 mmHg measured during left heart catheterisation supports diagnosis.
In Australia, RHC for HFpEF evaluation is performed at major tertiary centres with heart failure expertise. MBS item 38218 (right heart catheterisation) covers this procedure when clinically indicated. Exercise RHC requires specialised haemodynamic exercise equipment and is available at select centres including Royal Melbourne Hospital, Royal Prince Alfred Hospital, and the Alfred Hospital.
HFpEF Phenotyping
HFpEF is increasingly recognised as a syndrome with multiple distinct pathophysiological phenotypes, each with different treatment implications:
Investigations
Treatment Strategies
Management of HFpEF has evolved from a comorbidity-focused, symptom-driven approach to one incorporating disease-modifying therapies. The cornerstone of treatment is now SGLT2 inhibitors, supported by diuretic therapy, comorbidity management, and exercise-based rehabilitation.
SGLT2 Inhibitors — First-Line Disease-Modifying Therapy
SGLT2 inhibitors represent the most significant therapeutic advance in HFpEF management. Two landmark trials have established their efficacy:
Diuretic Management
Loop diuretics remain the mainstay of symptomatic management in HFpEF, addressing congestion through natriuresis and diuresis. They provide no mortality benefit but are essential for symptom control.
Comorbidity Treatment — Integrated Approach
HFpEF is rarely an isolated cardiac condition. Most patients have ≥3 comorbidities, and addressing these comprehensively is fundamental to improving outcomes. Key principles include:
- Aggressive blood pressure control — target <130/80 mmHg. ACE inhibitors, ARBs, and calcium channel blockers are all appropriate. ARNI (sacubitril/valsartan) showed numerical but non-significant benefit in PARAGON-HF (LVEF ≥57% subgroups and women showed possible benefit).
- Rate and rhythm control for atrial fibrillation — beta-blockers, digoxin, or calcium channel blockers for rate control. Catheter ablation may be superior to pharmacological rhythm control in HFpEF with AF (CASTLE-HFpEF subgroup analyses ongoing).
- Weight management — GLP-1 receptor agonists (semaglutide) for BMI ≥30 (see Emerging Therapies section). Multidisciplinary weight management programmes.
- Diabetes optimisation — SGLT2 inhibitors as first-line. GLP-1 RA for additional benefit. Avoid pioglitazone and saxagliptin (worsen HF). Metformin safe if eGFR >30.
- Iron replacement — IV ferric carboxymaltose (Ferinject®) if ferritin <100 or 100–299 µg/L with transferrin saturation <20%. Improves exercise capacity and quality of life (AFFIRM-AHF, IRONMAN data applicable).
- Sleep-disordered breathing — screening and treatment of obstructive sleep apnoea with CPAP. OSA is present in 50–75% of HFpEF patients.
Exercise Training
Exercise-based cardiac rehabilitation is a Class I recommendation (Level A evidence) for HFpEF and provides some of the most meaningful improvements in functional capacity and quality of life:
- Supervised aerobic exercise training (3–5 sessions per week, 30–60 minutes per session, moderate intensity 60–80% peak HR) improves peak VO₂ by approximately 1.5–2.0 mL/kg/min — a clinically meaningful improvement.
- Resistance training (2–3 sessions per week) is complementary and improves peripheral muscle strength and exercise tolerance.
- The Ex-DHF trial and subsequent meta-analyses confirmed improvements in NYHA class, 6-minute walk distance, and quality of life scores (Minnesota Living with Heart Failure Questionnaire).
- Cardiac Rehabilitation programme referral should occur for all HFpEF patients who are medically stable — MBS items 13550, 13560, and 13565 support cardiac rehabilitation in Australia.
- For those unable to access supervised programmes, structured home-based exercise with telehealth monitoring is a reasonable alternative.
Quick Reference — HFpEF Pharmacotherapy
Comorbidity Management
HFpEF is fundamentally a disease of comorbidity. The majority of patients present with ≥5 comorbid conditions, and the interaction between these conditions drives disease progression, symptom burden, and adverse outcomes. An integrated, multidisciplinary approach to comorbidity management is essential.
Atrial Fibrillation
AF and HFpEF share bidirectional pathophysiology — AF promotes atrial myopathy and impaired filling, while the stiff, hypertrophied left ventricle of HFpEF promotes atrial dilation and AF substrate. Approximately 40–60% of HFpEF patients have AF, and its presence worsens symptoms, exercise capacity, and prognosis.
Anticoagulation in HFpEF with AF: Direct oral anticoagulants (DOACs) are preferred over warfarin. Assess bleeding risk using HAS-BLED score. Use CHA₂DS₂-VASc score for stroke risk stratification — most HFpEF patients with AF have a score ≥2 and require anticoagulation. Apixaban (Eliquis® 5 mg BD, reduce to 2.5 mg BD if ≥2 of: age ≥80, weight ≤60 kg, Cr ≥133 µmol/L) or rivaroxaban (Xarelto® 20 mg daily with food, reduce to 15 mg if eGFR 15–49) are the preferred agents.
Hypertension
Hypertension is the most prevalent comorbidity in HFpEF (present in 60–90% of patients) and is a major driver of LVH, diastolic dysfunction, and disease progression. Aggressive blood pressure management is critical:
- Target: <130/80 mmHg (consistent with 2023 ESH and AHA/ACC guidelines). In elderly patients (>80 years), target SBP 130–139 mmHg to avoid excessive hypotension.
- First-line agents: ACE inhibitors (perindopril, ramipril) or ARBs (candesartan, valsartan). ARBs showed benefit in the TOPCAT trial (spironolactone subgroup analysis for Americas region).
- Second-line: Calcium channel blockers (amlodipine for additional afterload reduction). Thiazide-like diuretics (indapamide) for volume/pressure management.
- ARNI (sacubitril/valsartan): The PARAGON-HF trial narrowly missed its primary endpoint but demonstrated benefit in subgroups with LVEF <57% and in women (RR 0.73, 95% CI 0.59–0.90). Consider ARNI for patients with HFpEF and LVEF close to the lower end of normal, particularly women. PBS-listed for HFrEF; use for HFpEF requires private prescription or authority application.
- Mineralocorticoid receptor antagonists (MRAs): Spironolactone 25–50 mg daily — the TOPCAT trial showed reduction in HF hospitalisation in the Americas cohort (HR 0.82) but not the Russian/Georgian cohort. Consider for patients with persistent congestion despite SGLT2i and diuretics, monitoring potassium closely.
Obesity
Obesity (BMI ≥30) is present in 60–80% of HFpEF patients and is increasingly recognised as a central pathophysiological driver rather than merely a comorbidity. The "obesity-HFpEF" phenotype is characterised by epicardial adiposity, systemic inflammation, expanded plasma volume, and impaired cardiopulmonary reserve.
- Target weight reduction of ≥10% of body weight through lifestyle modification, pharmacotherapy, and consideration of bariatric surgery.
- Semaglutide 2.4 mg SC weekly (Wegovy®): The STEP-HFpEF trial demonstrated significant improvements in HF symptoms (Kansas City Cardiomyopathy Questionnaire), exercise capacity (6MWD), body weight (mean −13.3%), and CRP. The STEP-HFpEF DM trial confirmed similar benefits in patients with type 2 diabetes.
- Bariatric surgery: Consider for BMI ≥40 or BMI ≥35 with significant comorbidities. Associated with reversal of LVH, improvement in diastolic function, and reduction in HF hospitalisation.
- Structured dietary counselling (Mediterranean diet, sodium restriction <2 g/day) as adjunctive therapy.
Diabetes Mellitus
Type 2 diabetes is present in 40–45% of HFpEF patients and accelerates myocardial fibrosis, microvascular dysfunction, and cardiomyocyte hypertrophy through advanced glycation end-products (AGEs), oxidative stress, and insulin resistance.
- SGLT2 inhibitors are first-line — provide both glucose-lowering and HF-specific benefit. Empagliflozin and dapagliflozin are PBS-listed for type 2 diabetes (General Benefit).
- GLP-1 receptor agonists (semaglutide, liraglutide) provide weight reduction and cardiovascular benefit. Semaglutide 0.5–1 mg SC weekly (Ozempic®) is PBS-listed (Authority Required) for type 2 diabetes.
- Avoid saxagliptin — associated with increased HF hospitalisation (SAVOR-TIMI 53). Other DPP-4 inhibitors (linagliptin, sitagliptin) appear neutral.
- Metformin remains safe in HFpEF if eGFR >30 mL/min/1.73 m². Reduce dose if eGFR 30–45.
- HbA1c target: individualised, generally <53 mmol/mol (7%) for most patients; less stringent (<64 mmol/mol / 8%) in elderly or frail patients.
Emerging Therapies
HFpEF remains an area of intense research activity. After decades of negative trials, the SGLT2 inhibitor breakthrough has catalysed a new era of therapeutic development. Several novel agents and approaches are in advanced clinical testing.
GLP-1 Receptor Agonists for Obesity-Related HFpEF
The STEP-HFpEF programme represents a paradigm shift in recognising obesity as a treatable cause of HFpEF:
- STEP-HFpEF (semaglutide 2.4 mg weekly): Mean weight loss of 13.3% vs. 2.6% placebo. Significantly improved KCCQ clinical summary score (+7.8 points difference, p<0.001), 6MWD (+20.3 m, p=0.008), and CRP (−38.5% relative reduction). These are clinically meaningful improvements.
- STEP-HFpEF DM: Confirmed consistent benefits in the diabetes subgroup with similar magnitude of improvement.
- Cardiovascular outcome data (MACE, HF hospitalisation) from larger, longer trials are pending.
- In Australia, semaglutide (Wegovy® 2.4 mg) is approved by the TGA for weight management; Ozempic® (0.5–1 mg) is PBS-listed for type 2 diabetes. PBS listing for obesity without diabetes is not currently available.
Soluble Guanylate Cyclase (sGC) Stimulators
Vericiguat (Verquvo®), an sGC stimulator, addresses the nitric oxide–sGC–cGMP pathway that is impaired in HFpEF. The VITALITY-HFpEF trial (vericiguat vs. placebo) did not meet its primary endpoint (change in KCCQ physical limitation score or 6MWD). However, post-hoc analyses suggest potential benefit in specific subgroups. Vericiguat is currently PBS-listed only for HFrEF (after recent worsening HF event) and is not recommended for routine HFpEF management.
Novel Therapeutic Targets
| Therapy | Mechanism | Trial | Status |
|---|---|---|---|
| Finerenone | Non-steroidal MRA (more selective for MR, less hyperkalaemia) | FINEARTS-HF (2024) — met primary endpoint (HF events + CV death) | Positive. HR 0.84 (95% CI 0.74–0.95). Regulatory submission expected. Not yet TGA-approved for HFpEF. |
| Tirzepatide | Dual GIP/GLP-1 receptor agonist | SUMMIT trial (obesity + HFpEF) — results expected 2025 | Phase III. Anticipated to combine weight loss and metabolic benefits. Mounjaro® TGA-approved for T2DM. |
| Ziltivekimab | IL-6 monoclonal antibody (anti-inflammatory) | RESHAPE-HF2 (2024) — met primary endpoint | Positive. Targets the inflammatory phenotype. Not yet approved. |
| Tafamidis | TTR stabiliser (for ATTR cardiac amyloidosis) | ATTR-ACT — 30% reduction in all-cause mortality | PBS-listed (Authority Required) for ATTR cardiomyopathy. Vyndaqel® 80 mg daily. Paradigm-changing for this under-recognised HFpEF phenotype. |
| Sotatercept | Activin signalling inhibitor (TGF-β superfamily) | STELLAR (pulmonary arterial hypertension). Studied for HFpEF with elevated PAP. | FDA-approved for PAH. Potential HFpEF application under investigation. |
| Inclisiran | siRNA targeting PCSK9 (LDL-C reduction) | VICTORION-HEART — CV outcome data in broader population | PBS-listed for familial hypercholesterolaemia. Potential role in atherosclerotic HFpEF phenotype. |
Precision Medicine Approaches
The future of HFpEF management is moving towards phenotype-directed therapy. Key precision medicine approaches include:
- Machine learning–derived phenogroups: Cluster analyses from TOPCAT, RELAX, and NEAT-HFpEF datasets have identified 3–5 distinct phenogroups with differential treatment responses. These are not yet ready for clinical use but inform trial design.
- Multi-omics profiling: Proteomic (including high-sensitivity troponin, GDF-15, galectin-3), metabolomic, and genomic biomarker panels may enable risk stratification and treatment selection.
- Cardiac MRI-based tissue characterisation: T1 mapping, extracellular volume fraction (ECV), and late gadolinium enhancement patterns can identify specific myocardial pathologies (fibrosis, infiltration, inflammation) guiding targeted therapy.
- Wearable technology and remote monitoring: Pulmonary artery pressure monitoring (CardioMEMS — not yet widely available in Australia), implantable cardiac monitors, and consumer-grade wearables for activity, HR, and weight trending.
- Phenotype-specific trial design: Future trials are increasingly enrolling specific HFpEF phenotypes (e.g., obesity-predominant, AF-predominant) rather than the all-comer HFpEF population, which may have diluted treatment effects in earlier negative trials.
Special Populations
HFpEF management must be tailored to account for physiological differences, comorbidity patterns, and drug safety profiles in specific populations.
Pregnancy
Paediatrics
Elderly (≥75 years)
Renal Impairment
Hepatic Impairment
Immunocompromised
Monitoring
HFpEF requires longitudinal, structured monitoring to detect early decompensation, optimise therapy, and prevent hospitalisation. A multidisciplinary heart failure team approach (cardiologist, GP, heart failure nurse specialist, pharmacist, dietitian, exercise physiologist) is recommended.
Monitoring Schedule
Comprehensive baseline assessment: symptoms (NYHA class, KCCQ score), vital signs (BP, HR, weight, SpO₂), ECG, transthoracic echocardiography with diastolic function assessment, BNP/NT-proBNP, FBC, UEC, LFTs, HbA1c, fasting lipids, iron studies (ferritin, transferrin saturation), TFTs. Establish patient-specific self-management plan with daily weight monitoring.
Follow-up after initiating SGLT2 inhibitor or diuretic: renal function (eGFR, K⁺), volume status assessment, BP, weight, symptom review. Adjust diuretic dose based on clinical congestion. Ensure sick-day rules understood for SGLT2i.
Clinical review: symptom assessment (NYHA, KCCQ), weight, BP, HR, medication adherence, side-effect assessment. UEC, BNP/NT-proBNP every 3 months. Referral to cardiac rehabilitation if not already enrolled. Dietitian review for sodium restriction and weight management.
Repeat echocardiography to assess LV function, LA size, diastolic parameters, and RV function. Review and optimise comorbidity management (HbA1c, lipids, BP). Iron studies recheck if previously deficient. Functional capacity assessment (6MWT or CPET if available). Screen for AF (Holter monitor if palpitations or new symptoms).
Urgent review for: weight gain >2 kg in 3 days, worsening dyspnoea (NYHA class increase), new-onset orthopnoea or paroxysmal nocturnal dysnoea, peripheral oedema worsening, syncope or presyncope. Assess for precipitants (AF, infection, dietary indiscretion, medication non-adherence, ischaemia). Consider hospital admission if congestion not responsive to outpatient diuretic adjustment within 48 hours.
Self-Management Education
Patient self-management is a cornerstone of HFpEF care. Key elements include:
- Daily weight monitoring at the same time each morning (before breakfast, after voiding). Report gain of >1.5 kg in 24 hours or >2 kg in 3 days.
- Sodium restriction: target <2 g/day. Education on reading food labels, avoiding processed foods, and low-sodium cooking techniques.
- Fluid management: typically 1.5–2 L/day fluid intake unless severe hyponatraemia or diuretic resistance.
- Sick-day management rules for SGLT2 inhibitors — temporarily withhold if unable to maintain oral intake, vomiting, diarrhoea, or acutely unwell. Seek medical review within 24–48 hours.
- Medication adherence tools — medication blister packs (Webster packs), smartphone reminders, carer support.
- Recognition of decompensation symptoms and when to seek emergency care.
📚 References
- 1. Pieske B, Tschöpe C, de Boer RA, et al. How to diagnose heart failure with preserved ejection fraction: the HFA-PEFF diagnostic algorithm: a consensus recommendation from the Heart Failure Association (HFA) of the European Society of Cardiology (ESC). Eur Heart J. 2019;40(40):3297-3317.
- 2. Anker SD, Butler J, Filippatos G, et al. Empagliflozin in heart failure with a preserved ejection fraction. N Engl J Med. 2021;385(16):1451-1461. (EMPEROR-Preserved)
- 3. Solomon SD, McMurray JJV, Claggett B, et al. Dapagliflozin in heart failure with mildly reduced or preserved ejection fraction. N Engl J Med. 2022;387(12):1089-1098. (DELIVER)
- 4. Kosiborod MN, Abildstrøm SZ, Borlaug BA, et al. Semaglutide in patients with heart failure with preserved ejection fraction and obesity. N Engl J Med. 2023;389(12):1069-1084. (STEP-HFpEF)
- 5. Pitt B, Pfeffer MA, Assmann SF, et al. Spironolactone for heart failure with preserved ejection fraction. N Engl J Med. 2014;370(15):1383-1392. (TOPCAT)
- 6. Solomon SD, McMurray JJV, Anand IS, et al. Angiotensin-neprilysin inhibition in heart failure with preserved ejection fraction. N Engl J Med. 2019;381(17):1609-1620. (PARAGON-HF)
- 7. McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42(36):3599-3726.
- 8. Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the management of heart failure. Circulation. 2022;145(18):e895-e1032.
- 9. Australian Institute of Health and Welfare. Heart failure in Australia. AIHW, Canberra. 2023.
- 10. National Heart Foundation of Australia and Cardiac Society of Australia and New Zealand. Guidelines for the prevention, detection, and management of heart failure in Australia. 2024 Update.
- 11. Borlaug BA, Anstrom KJ, Lewis GD, et al. Effect of inorganic nitrite vs placebo on exercise capacity among patients with heart failure with preserved ejection fraction: the INDIE-HFpEF randomized clinical trial. JAMA. 2018;320(17):1764-1773.
- 12. Redfield MM, Chen HH, Borlaug BA, et al. Effect of phosphodiesterase-5 inhibition on exercise capacity and clinical status in heart failure with preserved ejection fraction: a randomized clinical trial. JAMA. 2013;309(12):1268-1277. (RELAX)
- 13. Nagueh SF, Smiseth OA, Appleton CP, et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2016;29(4):277-314.
- 14. RHDAustralia (RHD Australia), National Heart Foundation of Australia and Cardiac Society of Australia and New Zealand. Australian guideline for prevention, diagnosis, and management of acute rheumatic fever and rheumatic heart disease. 3rd ed. 2020.
- 15. Maurer MS, Schwartz JH, Gundapaneni B, et al. Tafamidis treatment for patients with transthyretin amyloid cardiomyopathy. N Engl J Med. 2018;379(11):1007-1016. (ATTR-ACT)
- 16. Mentz RJ, Kelly JP, von Lueder TG, et al. Noncardiac comorbidities in heart failure with preserved versus reduced ejection fraction. J Am Coll Cardiol. 2014;64(21):2261-2269.
- 17. Shah SJ, Borlaug BA, Kitzman DW, et al. Research priorities for heart failure with preserved ejection fraction: National Heart, Lung, and Blood Institute Working Group summary. Circulation. 2020;141(12):1001-1026.
- 18. Bhagra SK, Pettit S, Parameshwar J. Cardiac transplantation: indications, eligibility and current outcomes. Heart. 2019;105(3):252-260.