📋 Key Information Summary
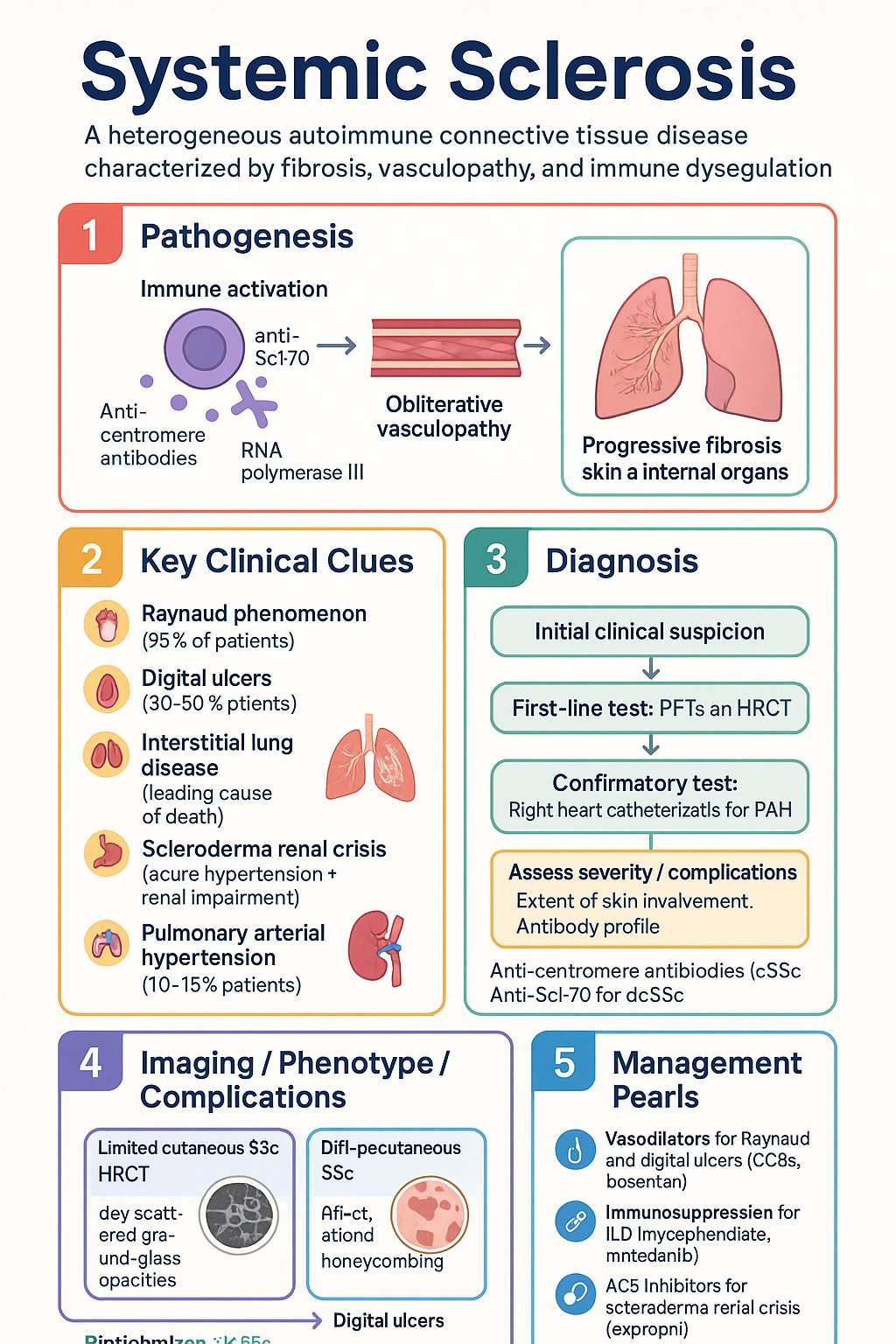
- Systemic sclerosis (SSc) is a heterogeneous autoimmune connective tissue disease characterised by fibrosis, vasculopathy, and immune dysregulation.
- Classified as limited cutaneous SSc (lcSSc) or diffuse cutaneous SSc (dcSSc) based on extent of skin involvement; dcSSc carries higher risk of internal organ disease.
- Anti-centromere antibodies (ACA) associate with lcSSc and PAH; anti-Scl-70 (topoisomerase I) and RNA polymerase III associate with dcSSc and ILD/scleroderma renal crisis respectively.
- Raynaud phenomenon is present in >95% of patients; secondary Raynaud with nailfold capillaroscopy abnormalities predicts internal organ involvement.
- Digital ulcers occur in 30–50% of patients; treat with vasodilators (CCBs, bosentan, iloprost) and wound care.
- Interstitial lung disease (ILD) is the leading cause of death in SSc; screen with PFTs and HRCT; treat with immunosuppression (mycophenolate preferred) ± nintedanib.
- Scleroderma renal crisis (SRC) — acute hypertension + renal impairment — is an emergency. ACE inhibitors (captopril) are first-line; avoid corticosteroids >15 mg/day prednisolone.
- Pulmonary arterial hypertension (PAH) affects 10–15% of SSc patients; screen annually with echocardiography and PFTs; confirm with right heart catheterisation.
- GI dysmotility affects >90% of patients; treat symptomatically with prokinetics, PPI, antibiotics for bacterial overgrowth, and nutritional support.
- Aboriginal and Torres Strait Islander patients may have reduced access to specialist rheumatology and pulmonary services; telehealth and outreach clinics are essential.
Introduction & Australian Epidemiology
Systemic sclerosis (SSc) is a chronic multisystem autoimmune disease characterised by immune activation, obliterative vasculopathy, and progressive fibrosis of skin and internal organs. It remains one of the most lethal rheumatic diseases, with standardised mortality ratios of 3–5× the general population.
| Parameter | Data |
|---|---|
| Australian prevalence | ~20 per 100,000 adults |
| Annual incidence | ~2 per 100,000 |
| Female : male ratio | 4 : 1 |
| Peak onset | 30–50 years |
| Leading causes of death | ILD/fibrosis, PAH, cardiac involvement |
Australian data from the Scleroderma Australia Registry show that dcSSc accounts for ~40% of cases, with higher mortality compared to lcSSc. The disease carries substantial morbidity from vascular, pulmonary, and gastrointestinal complications.
Limited vs Diffuse Cutaneous SSc
Classification by LeRoy criteria determines prognosis and organ risk stratification.
| Feature | lcSSc | dcSSc |
|---|---|---|
| Skin fibrosis | Distal to elbows/knees, face | Proximal (trunk, upper arms, thighs) |
| Antibodies | ACA (70–80%) | Scl-70 (30%), RNA Pol III (20%) |
| PAH risk | Higher | Lower |
| ILD risk | Moderate | High (~80%) |
| SRC risk | Low (~2%) | Higher (~15%) |
| Natural history | Slowly progressive | Rapid skin progression (peak 3–5 yr), then plateau |
| 10-year survival | ~75% | ~55–65% |
Raynaud Phenomenon & Digital Ulcers
Raynaud phenomenon (RP) is present in >95% of SSc patients and is often the earliest manifestation. Secondary RP is distinguished by nailfold capillary abnormalities, specific autoantibodies, and tissue injury.
Management of Raynaud Phenomenon
Digital Ulcer Management — Step-Up Approach
Interstitial Lung Disease (SSc-ILD)
SSc-ILD is the leading cause of SSc-related death, present in up to 80% of dcSSc and 35% of lcSSc patients. NSIP is the predominant histological pattern.
Screening Protocol
- Baseline HRCT + PFTs (FVC, DLCO) at diagnosis
- Annual PFTs (FVC, DLCO) — more frequent if dcSSc or positive Scl-70
- Repeat HRCT if FVC decline >10% or new respiratory symptoms
- Consider screening with anti-Scl-70, anti-U3 RNP, and extent of skin disease
Treatment — Severity-Stratified
Scleroderma Renal Crisis (SRC)
Risk Factors
- Diffuse cutaneous disease, rapidly progressive skin thickening
- RNA polymerase III antibody positive
- New anaemia, thrombocytopenia, or pericardial effusion
- Corticosteroids >15 mg/day prednisolone — major trigger
- Cyclosporine, tacrolimus use
Emergency Management
Monitor BP, renal function, FBC (MAHA), LDH, haptoglobin closely. Nephrology referral for all SRC patients.

Pulmonary Arterial Hypertension (PAH)
PAH (mean PAP >20 mmHg, PAWP ≤15 mmHg, PVR >3 WU on RHC) affects 10–15% of SSc patients and is the second leading cause of death. Higher risk with lcSSc, ACA positivity, longstanding disease, and digital ulcer history.
Screening Protocol
- Annual echocardiography (TR velocity, RV function) + DLCO from diagnosis
- Symptom assessment: dyspnoea (WHO functional class), exercise tolerance
- Right heart catheterisation (RHC) if echocardiographic or functional concern
- DLCO <60% predicted or declining >15% — refer for RHC
PAH Therapy — Australian Access
| Drug class | Example | Route | PBS |
|---|---|---|---|
| ERA | Ambrisentan 5–10 mg daily | PO | Authority |
| PDE-5i | Sildenafil 20 mg TDS | PO | Authority |
| Prostacyclin | Treprostinil SC, epoprostenol IV | SC/IV | Authority |
| sGC stimulator | Riociguat 1–2.5 mg TDS | PO | Authority |
| Combination | ERA + PDE-5i (standard of care) | PO | Authority |
All SSc-PAH patients require referral to a designated PAH centre for RHC confirmation, treatment initiation, and ongoing monitoring. Anticoagulation is not routinely recommended in SSc-PAH (unlike idiopathic PAH).
GI Dysmotility
GI involvement affects >90% of SSc patients and significantly impairs quality of life. Fibrosis and vasculopathy cause smooth muscle atrophy and dysmotility from mouth to anus.
Site-Specific Manifestations & Treatment
| Site | Manifestation | Treatment |
|---|---|---|
| Oesophagus | GORD, dysphagia, Barrett's | PPI (omeprazole 20–40 mg daily), elevation of HOB, prokinetics |
| Stomach | Gastroparesis, early satiety | Metoclopramide 10 mg TDS or domperidone 10 mg TDS pre-meals |
| Small bowel | Bacterial overgrowth, pseudo-obstruction | Rotating antibiotics (ciprofloxacin, doxycycline, amoxicillin-clavulanate 1 week/month) |
| Colon | Constipation, pseudo-obstruction | Osmotic laxatives, fibre, prucalopride |
| Anorectal | Faecal incontinence | Biofeedback, sacral nerve stimulation if severe |
Special Populations
Aboriginal and Torres Strait Islander Health Considerations
📚 References
- 1. Denton CP, Khanna D. Systemic sclerosis. Lancet. 2017;390(10103):1685-1699.
- 2. LeRoy EC, Medsger TA. Criteria for the classification of early systemic sclerosis. J Rheumatol. 2001;28(7):1573-1576.
- 3. Kowal-Bielecka O, Fransen J, Avouac J, et al. Update of EULAR recommendations for the treatment of systemic sclerosis. Ann Rheum Dis. 2017;76(8):1327-1339.
- 4. Proudman SM, Stevens WM, Sahhar J, Englert H. Systemic sclerosis in Australia. Intern Med J. 2007;37(1):1-8.
- 5. Tashkin DP, Elashoff R, Clements PJ, et al. Cyclophosphamide versus placebo in scleroderma lung disease. N Engl J Med. 2006;354(25):2655-2666.
- 6. Distler O, Highland KB, Gahlemann M, et al. Nintedanib for systemic sclerosis-associated interstitial lung disease. N Engl J Med. 2019;380(26):2518-2528.
- 7. Tynan MJ, Tay LK, MacGregor L, et al. Mycophenolate mofetil for scleroderma-associated interstitial lung disease. Rheumatology. 2021;60(5):2325-2334.
- 8. Guillevin L, Bérezné A, Seror R, et al. Scleroderma renal crisis: a retrospective multicentre study on 91 patients and 427 controls. Rheumatology. 2012;51(3):460-467.
- 9. Galiè N, Humbert M, Vachiery JL, et al. 2015 ESC/ERS guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Heart J. 2016;37(1):67-119.
- 10. McMahan ZH, Wigley FM. Gastrointestinal tract involvement in systemic sclerosis. Curr Opin Rheumatol. 2014;26(6):637-642.
- 11. Australian Institute of Health and Welfare (AIHW). Aboriginal and Torres Strait Islander health performance framework. Canberra: AIHW; 2023.
- 12. Herrick AL, Muir L, Munir N, et al. Bosentan for the treatment of digital ulcers in patients with systemic sclerosis: pooled analysis of two randomised controlled trials. Arthritis Res Ther. 2013;15(6):R225.
- 13. Ghofrani HA, Galiè N, Grimminger F, et al. Riociguat for the treatment of pulmonary arterial hypertension. N Engl J Med. 2013;369(4):330-340.
- 14. National Health and Medical Research Council (NHMRC). Australian guidelines to reduce health risks from drinking alcohol. Canberra: NHMRC; 2020.
- 15. Sahhar J, Littlejohn G, Conron M. Fibrosing alveolitis in systemic sclerosis: the need for early screening and treatment. Intern Med J. 2004;34(11):620-624.