📋 Key Information Summary
- Kawasaki disease (KD) is the most common cause of acquired heart disease in children in developed countries, including Australia.
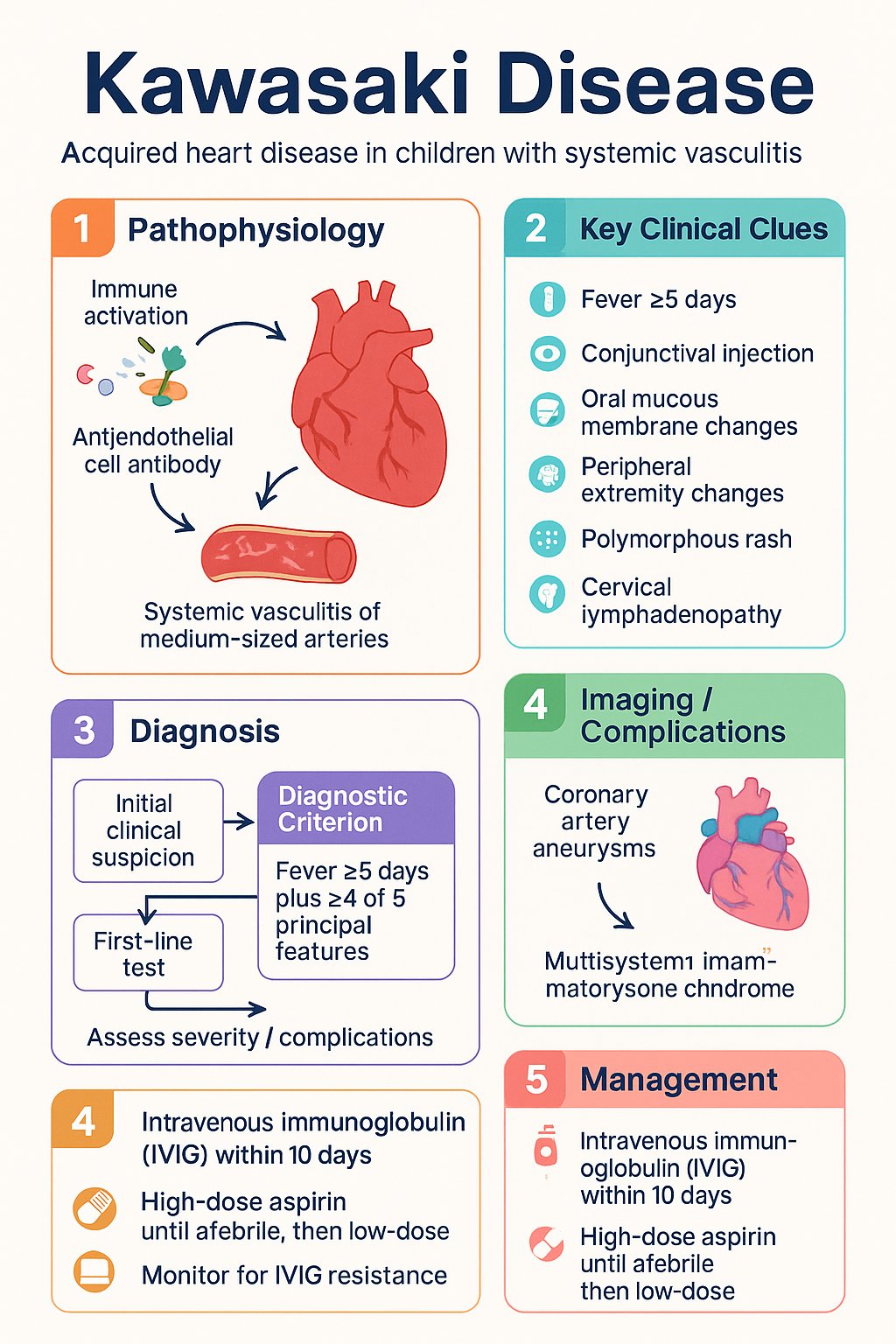
- Diagnosis is clinical: fever ≥5 days plus ≥4 of 5 principal features (conjunctival injection, oral mucous membrane changes, peripheral extremity changes, polymorphous rash, cervical lymphadenopathy).
- Incomplete KD should be considered in any child with prolonged unexplained fever and fewer than 4 principal features, using specific laboratory and echocardiographic criteria.
- Coronary artery aneurysms (CAA) occur in ~25% of untreated children; risk is highest in infants <1 year and males.
- First-line treatment is intravenous immunoglobulin (IVIG) 2 g/kg single infusion PLUS high-dose aspirin (80-100 mg/kg/day) until afebrile, then low-dose aspirin (3-5 mg/kg/day).
- Treatment must be initiated within the first 10 days of illness, ideally within the first 7 days, to maximise efficacy in preventing CAA.
- ~10-20% of patients are IVIG-resistant (persistent/recurrent fever 36h-7 days after initial IVIG); second-line options include a second IVIG dose, infliximab, or corticosteroids.
- Atypical or incomplete KD is common and requires a high index of suspicion; the same treatment principles apply.
- Long-term cardiac follow-up risk-stratified based on coronary artery status, from no follow-up for normal arteries to lifelong cardiology for giant aneurysms.
- Multisystem Inflammatory Syndrome in Children (MIS-C) shares features with KD but is a distinct post-COVID-19 hyperinflammatory condition with different epidemiology, cardiac involvement patterns, and treatment responses.
- Aboriginal and Torres Strait Islander children may have a higher incidence and/or different presentation patterns; culturally safe care and access to echocardiography are critical.
Introduction & Australian Epidemiology
Kawasaki disease (KD) is an acute, self-limiting systemic vasculitis of medium-sized arteries, predominantly affecting the coronary arteries. It is the leading cause of acquired heart disease in children in Australia and other developed nations, surpassing acute rheumatic fever.
In Australia, the annual incidence is estimated at 8-10 per 100,000 children under 5 years, though rates may be higher in some populations. There is a marked seasonal variation, with peaks in winter and spring. The disease has a strong predilection for young children: 80% of cases occur in children under 5 years, with a peak incidence at 18-24 months. Boys are affected 1.5 times more frequently than girls.
Diagnostic Criteria
The diagnosis of classic (complete) Kawasaki disease is clinical. There is no specific diagnostic test. The American Heart Association (AHA) criteria are used in Australia.
Classic (Complete) Kawasaki Disease
Fever persisting for at least 5 days* PLUS at least 4 of the following 5 principal features:
| Principal Feature | Typical Characteristics |
|---|---|
| 1. Bilateral non-exudative conjunctival injection | Bulbar conjunctivitis, limbic sparing, painless, usually begins shortly after fever onset. |
| 2. Changes of the oral mucous membranes | Erythema and cracking of lips, strawberry tongue, diffuse oropharyngeal erythema. |
| 3. Changes of the peripheral extremities | Acute: Erythema and oedema of hands and feet. Subacute (weeks 2-3): Periungual and finger/toe tip peeling. |
| 4. Polymorphous rash | Maculopapular, diffuse erythroderma, erythema multiforme-like; typically trunk and extremities; no vesicles or crusts. |
| 5. Cervical lymphadenopathy | ≥1.5 cm diameter, usually unilateral, non-suppurative. Least common feature (~50-75% of cases), but may be the presenting sign. |
*Fever can be less than 5 days if principal features are present and other causes excluded.
Incomplete Kawasaki Disease
This term applies to patients with persistent unexplained fever and fewer than 4 principal features. It is particularly common in infants <6 months, who are at highest risk for CAA. Diagnosis requires a high index of suspicion and use of the AHA algorithm involving laboratory markers (CRP, ESR, albumin, ALT, platelets, WCC, urine leucocytes) and echocardiography.
Diagnostic Algorithm for Suspected Incomplete KD:
Coronary Artery Aneurysm Risk
Coronary artery aneurysms (CAA) are the most serious complication. In the pre-IVIG era, ~25% of children developed CAA. With timely IVIG treatment, this risk is reduced to 3-5%. Giant aneurysms (z-score ≥10 or diameter >8 mm) carry the highest risk of thrombosis, stenosis, myocardial infarction, and death.
Risk Factors for CAA Development:
- Male sex (1.5x higher risk)
- Age <1 year (especially <6 months) - often present later, with incomplete features.
- Late treatment (after day 10 of illness).
- IVIG resistance (persistent fever after initial treatment).
- High baseline inflammatory markers (CRP, ESR) and low albumin.
- Low haemoglobin, low sodium at presentation.
Coronary Artery Assessment & Z-Scores:
Echocardiography is the primary imaging modality. Coronary arteries are measured and compared to body surface area to generate a z-score (standard deviation from the mean). This provides objective criteria for classification:
IVIG & Aspirin Therapy
Standard first-line therapy is a single high-dose infusion of intravenous immunoglobulin (IVIG) combined with high-dose aspirin.
Aspirin Use: Australian Context
While the role of high-dose aspirin in reducing CAA risk is debated internationally, it remains standard practice in Australia per national guidelines. The primary anti-inflammatory effect is from IVIG. The low-dose aspirin phase provides antiplatelet activity. Reye's syndrome risk: Parents must be counselled about the signs of Reye's and to avoid use of over-the-counter NSAIDs or aspirin-containing products for other febrile illnesses during treatment.
Refractory & Atypical Kawasaki Disease
IVIG-Resistant (Refractory) Kawasaki Disease
Defined as persistent or recrudescent fever 36 hours to 7 days after completion of the first IVIG infusion. This occurs in 10-20% of patients and is a significant risk factor for CAA. Risk scoring (e.g., Kobayashi, Egami, Sano scores) can identify high-risk patients but are not universally adopted.
Second-Line Treatment Options (Typically in consultation with tertiary paediatric cardiology/rheumatology):
Atypical Kawasaki Disease
Atypical KD refers to patients who present with supplemental features not part of the classic criteria (e.g., arthritis, uveitis, meningitis, shock, macrophage activation syndrome) in addition to fever and fewer than 4 principal features. It is a subset of incomplete KD. The threshold for treatment should be low, as these patients are at high risk for CAA.

Long-Term Cardiac Follow-Up
Follow-up is risk-stratified based on the worst-ever coronary artery z-score. Management is coordinated by a paediatric cardiologist.
| Risk Level | Coronary Status | Recommended Follow-Up & Therapy |
|---|---|---|
| Level I (No involvement) | Z-score always <2.0 | Discharge from cardiology after 6-8 week echo. No long-term restrictions. Cardiovascular risk factor counselling (healthy lifestyle). |
| Level II (Dilation only) | Z-score 2.0 to <2.5 | Low-dose aspirin until regression confirmed. Repeat echo at 1 year. May discharge if regression complete. |
| Level III (Small aneurysm) | Z-score ≥2.5 to <5 | Low-dose aspirin long-term. Annual cardiology review with echo. Stress testing at transition to adult care. |
| Level IV (Medium/Giant aneurysm) | Z-score ≥5 | Low-dose aspirin + anticoagulation (e.g., warfarin INR 2.0-2.5, or DOAC). 6-12 monthly cardiology. Stress testing. Consider CT/MR angiography. Lifelong follow-up required. |
Transition to Adult Care
Adolescents with persistent aneurysms require a structured transition to an adult congenital heart disease cardiologist. Key issues include ongoing anti-thrombotic therapy, pregnancy counselling (especially for women on warfarin), and lifelong monitoring for ischaemic events.
MIS-C Overlap & Differentiation
Multisystem Inflammatory Syndrome in Children (MIS-C) is a post-infectious hyperinflammatory condition temporally associated with SARS-CoV-2 infection. It shares significant clinical overlap with KD but is now considered a distinct entity.
| Feature | Classic Kawasaki Disease | Typical MIS-C |
|---|---|---|
| Epidemiology | Peak <5 years, Asian ancestry higher incidence. | School-age (median ~8 years), often previously well. All ethnicities. |
| Serology | Not linked to COVID-19. | SARS-CoV-2 PCR or antibody positive in ~80-90%. |
| Presenting Features | Fever, classic mucocutaneous features. | Fever, severe abdominal pain, vomiting, diarrhoea, shock, myocardial dysfunction. |
| Cardiac | Coronary artery aneurysms (medium vessel). | Myocarditis, ventricular dysfunction, valvulitis, coronary dilation (usually mild). |
| Labs | High CRP/ESR, thrombocytosis later. | Very high CRP/Ferritin/D-dimer, lymphopenia, thrombocytopenia, elevated troponin/BNP. |
| Treatment Response | IVIG is first-line and effective. | IVIG alone often insufficient. Often requires IVIG + corticosteroids ± biologics (Anakinra, infliximab). |
📚 References
- 1. McCrindle BW, Rowley AH, Newburger JW, et al. Diagnosis, Treatment, and Long-Term Management of Kawasaki Disease: A Scientific Statement for Health Professionals From the American Heart Association. Circulation. 2017;135(17):e927-e999.
- 2. > Newburger JW, Takahashi M, Burns JC. Kawasaki Disease. J Am Coll Cardiol. 2016;67(14):1738-1749.
- 3. Australian Institute of Health and Welfare (AIHW). Kawasaki disease in Australia. Cat. no. CVD 90. Canberra: AIHW; 2021.
- 4. RACGP. Kawasaki disease. In: Royal Australian College of General Practitioners (RACGP) clinical resources. East Melbourne, Vic: RACGP.
- 5. Children's Health Queensland. Kawasaki Disease: Clinical Practice Guideline. Brisbane: CHQ; 2022.
- 6. Dionne A, Burns JC, Dahdah N, et al. Treatment Intensification in Patients With Kawasaki Disease and Coronary Aneurysm at Diagnosis. Pediatrics. 2019;143(6):e20183341.
- 7. Henderson D, Bakshi A, Shah V, et al. The Australian experience with multisystem inflammatory syndrome in children (MIS-C) during the COVID-19 pandemic. Med J Aust. 2021;215(6):261-265.
- 8. Saundankar J, Yim D, Itotoh B, et al. The epidemiology and clinical features of Kawasaki disease in Australia. Pediatrics. 2014;133(4):e1009-e1014.
- 9. Manlhiot C, Millar K, Golding F, McCrindle BW. Improved classification of coronary artery abnormalities after Kawasaki disease. J Am Coll Cardiol. 2010;56(14):1121-1128.
- 10. > Galeotti C, Kaveri SV, Cimaz R, et al. Predisposing factors, pathogenesis, and management of Kawasaki disease-related coronary artery vasculitis. J Inflamm Res. 2021;14:1409-1425.
- 11. National COVID-19 Clinical Evidence Taskforce. Caring for people with COVID-19: Multisystem inflammatory syndrome in children (MIS-C). Melbourne; 2022.
- 12. RHDAustralia (Australian Government Department of Health). Kawasaki Disease. In: Australian Guideline for Prevention, Diagnosis and Management of Acute Rheumatic Fever and Rheumatic Heart Disease (3rd edition). Darwin: RHDAustralia; 2020.