📋 Key Information Summary
- Psoriatic arthritis (PsA) is a chronic, immune-mediated inflammatory arthritis associated with psoriasis, affecting joints, entheses, tendons, and skin.
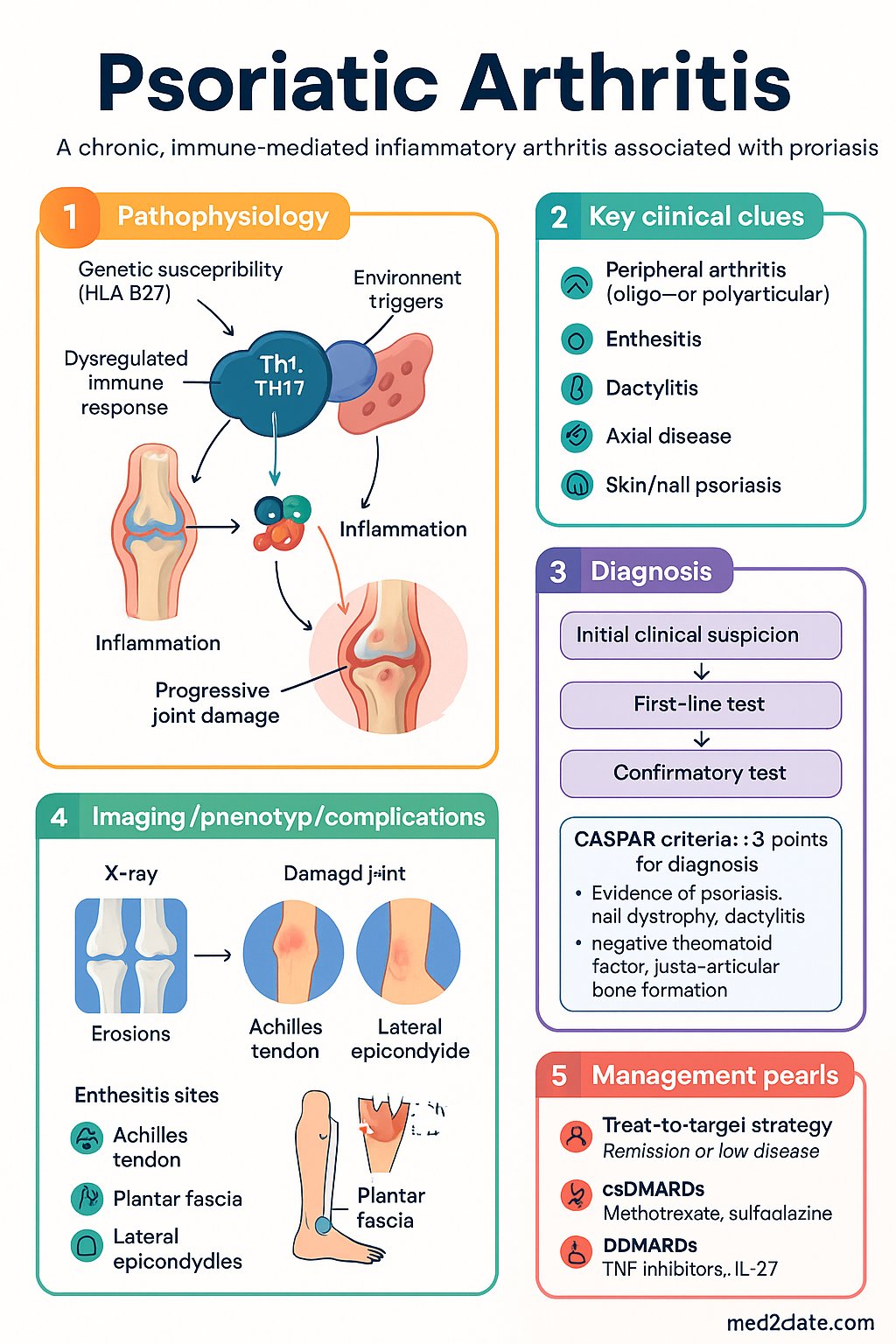
- Diagnosis is clinical, supported by the CASPAR classification criteria; no single definitive test exists.
- Key clinical domains: peripheral arthritis (oligo- or polyarticular), enthesitis, dactylitis, axial disease, and skin/nail psoriasis.
- Treatment target is remission or low disease activity, using a treat-to-target strategy with regular monitoring (e.g., DAPSA, PASDAS).
- First-line for peripheral arthritis: csDMARDs like methotrexate (PBS-listed). Consider sulfasalazine or leflunomide.
- For severe/refractory disease, or predominant enthesitis/dactylitis, advance to biologic DMARDs (bDMARDs): TNF inhibitors (e.g., adalimumab), IL-17 inhibitors (e.g., secukinumab), or IL-23 inhibitors (e.g., guselkumab).
- JAK inhibitors (e.g., tofacitinib) are an alternative oral option for bDMARD-inadequate response, with specific PBS Authority criteria.
- Nail disease and enthesitis often require targeted therapy; topical therapies alone are insufficient for significant PsA.
- Screen for cardiovascular risk, metabolic syndrome, and depression—common comorbidities.
- Mandatory assessment and management of Aboriginal and Torres Strait Islander patients, addressing barriers to specialist access and culturally safe care.
- Live vaccines are contraindicated in patients on biologics/JAK inhibitors.
Introduction & Australian Epidemiology
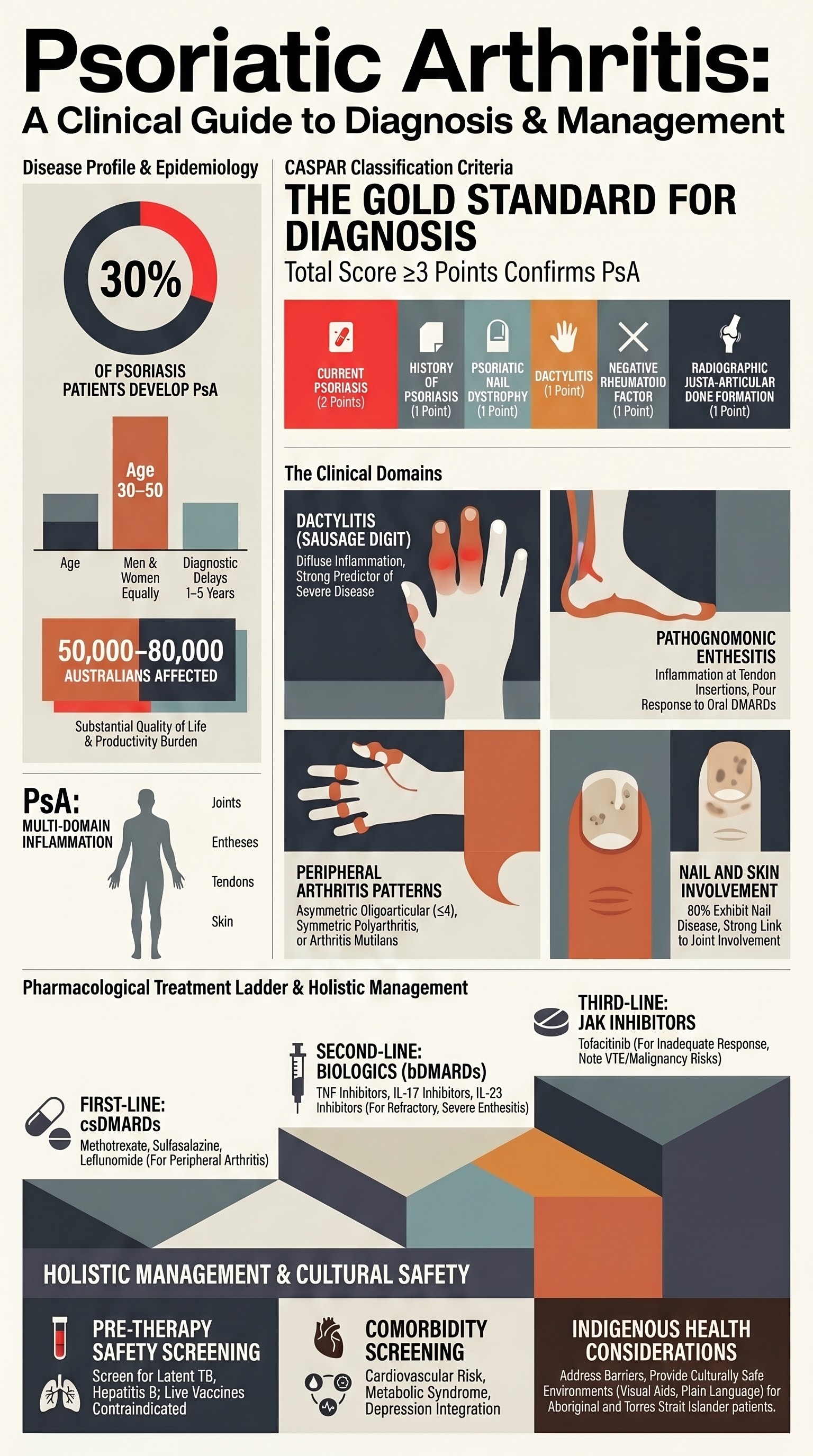
Psoriatic arthritis (PsA) is a distinct inflammatory arthritis occurring in up to 30% of individuals with psoriasis. It is classified as a spondyloarthropathy and is characterised by inflammation of joints, entheses, tendons, and skin. In Australia, the prevalence of PsA is estimated at 0.2–0.3% of the general population, affecting approximately 50,000–80,000 Australians. It presents equally in men and women, typically between the ages of 30 and 50. A significant diagnostic delay of 1–5 years is common. The disease burden is substantial, with impact on quality of life, physical function, and work productivity comparable to rheumatoid arthritis.
The pathogenesis involves a complex interplay of genetic susceptibility (strong association with HLA-B27 and other HLA alleles), environmental triggers, and dysregulated immune responses involving Th1, Th17, and IL-23 pathways. This leads to inflammation, enthesitis, and progressive joint damage.
CASPAR Classification Criteria
The Classification Criteria for Psoriatic Arthritis (CASPAR) are the standard for diagnosis in clinical practice and trials. They have a specificity of ~99% and sensitivity of ~91%.
| Criterion | Points |
|---|---|
| Evidence of psoriasis (current, history, or family) Current psoriasis: 2 points Psoriasis history or family history: 1 point (If current psoriasis, do not score history/family) |
2 or 1 |
| Psoriatic nail dystrophy (onycholysis, pitting, hyperkeratosis) | 1 |
| Dactylitis (swelling of entire digit, current or history) | 1 |
| Negative rheumatoid factor (by any method except ELISA) | 1 |
| Juxta-articular bone formation on X-ray (excluding osteophytes) | 1 |
Peripheral Arthritis
Peripheral joint involvement is the hallmark of PsA. Patterns include:
- Asymmetric oligoarticular: Most common at presentation (70%). Affects ≤4 joints, often large joints (knee, ankle) and DIP joints.
- Symmetric polyarthritis: Resembles rheumatoid arthritis (RA) but often with fewer joints and concurrent DIP involvement.
- Distal interphalangeal (DIP) predominant: Classic pattern, strongly associated with nail disease.
- Arthritis mutilans: Severe, deforming form with osteolysis ("pencil-in-cup" deformity, telescoping digits). Rare (5%).
Initial assessment should include joint count (68/66 tender/swollen), patient global VAS, and function (HAQ-DI). Baseline X-rays of hands and feet are essential for detecting erosions, joint space narrowing, and new bone formation.
Enthesitis
Inflammation at the insertion of tendons and ligaments into bone is a pathognomonic feature of PsA and other spondyloarthropathies. Common sites include:
- Achilles tendon insertion and plantar fascia (calcaneus)
- Lateral epicondyles of the elbow
- Medial femoral condyles
- Supraspinatus insertion at the greater tuberosity
Clinical examination is key; tenderness on palpation is the primary finding. Ultrasound or MRI can be used for confirmation but are not always necessary. The Leeds Enthesitis Index (LEI) or Maastricht Ankylosing Spondylitis Enthesitis Score (MASES) are used for monitoring.
Dactylitis
Also known as "sausage digit," dactylitis is diffuse inflammation of an entire digit (finger or toe), involving joint, tendon sheath, and soft tissue. It is highly characteristic of PsA (present in up to 50% of patients).
Dactylitis is associated with a higher risk of radiographic progression and is a strong predictor of more severe disease. It is graded as present/absent or by the number of affected digits.
Treatment considerations are similar to enthesitis: it is a poor prognostic factor and may require escalation to bDMARDs earlier than peripheral arthritis alone.
Skin & Nail Disease
The severity of skin psoriasis does not correlate with the severity of joint disease. However, nail involvement is strongly linked.
- Nail psoriasis: Pitting, onycholysis, subungual hyperkeratosis, and splinter haemorrhages. Present in ~80% of PsA patients vs. ~40% of those with cutaneous psoriasis alone.
- Skin: Plaque psoriasis is most common. Guttate, inverse, or pustular variants may occur.
Integrated management with dermatology is often required. Systemic therapies that treat both domains (e.g., methotrexate, biologics) are preferred. Topical therapies (corticosteroids, vitamin D analogues) are for skin-limited disease.
MBS Item 110: A GP Management Plan (GPMP) can be initiated for chronic condition management.
Biologic & Targeted Synthetic DMARD Therapy
Therapy escalation is based on disease activity, prognostic factors (e.g., high CRP, erosions, enthesitis, dactylitis), and patient preference. The Pharmaceutical Benefits Scheme (PBS) has specific Authority criteria for biologic and targeted synthetic DMARDs (b/tsDMARDs).
TNF Inhibitors (TNFi)
First-line biologics. All require failure of at least one csDMARD.
IL-17 Inhibitors
Effective for skin, enthesitis, and arthritis. An option after TNFi failure or as first-line.
IL-23 Inhibitors
Primarily indicated for psoriasis; evidence for PsA is robust for some agents. Generally used after TNFi or IL-17i.
JAK Inhibitors
Oral targeted therapy. For patients who have had an inadequate response or intolerance to a TNF inhibitor. PBS criteria are strict.
Aboriginal and Torres Strait Islander Health
The prevalence and severity of autoimmune conditions, including PsA, in Aboriginal and Torres Strait Islander peoples are not well-documented but are likely under-diagnosed. Significant barriers to care exist.
📚 References
- 1. Coates LC, Helliwell PS. Psoriatic arthritis: state of the art review. Clin Med (Lond). 2017;17(1):65-70.
- 2. Taylor W, Gladman D, Helliwell P, et al. Classification criteria for psoriatic arthritis: development of new criteria from a large international study. Arthritis Rheum. 2006;54(8):2665-73.
- 3. Smolen JS, Schöls M, Braun J, et al. Treating axial spondyloarthritis and peripheral spondyloarthritis, especially psoriatic arthritis, to target: 2017 update of recommendations by an international task force. Ann Rheum Dis. 2018;77(1):3-17.
- 4. Gossec L, Baraliakos X, Kerschbaumer A, et al. EULAR recommendations for the management of psoriatic arthritis with pharmacological therapies: 2019 update. Ann Rheum Dis. 2020;79(6):700-712.
- 5. Australian Government Department of Health. Pharmaceutical Benefits Scheme (PBS). Available at: https://www.pbs.gov.au. [Accessed November 2024] – for current Authority Required criteria.
- 6. Ritchlin CT, Colbert RA, Gladman DD. Psoriatic Arthritis. N Engl J Med. 2017;376(10):957-970.
- 7. Australian Institute of Health and Welfare (AIHW). Aboriginal and Torres Strait Islander health performance framework. 2023.
- 8. National Aboriginal Community Controlled Health Organisation (NACCHO). Cultural safety standards. 2022.
- 9. Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80(4):1029-1072.
- 10. Gladman DD, Ritchlin C. Clinical manifestations and diagnosis of psoriatic arthritis. In: UpToDate, Post TW (Ed), UpToDate, Waltham, MA. (Accessed November 2024).